
Molecular Geometry and VSEPR Theory Quiz
Interactive Video
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Hard

Jennifer Brown
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What does VSEPR Theory help us understand about molecules?
The color of the molecule
The geometric arrangement of electron groups
The speed of molecular reactions
The weight of the molecule
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
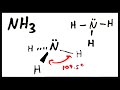
What is the molecular geometry of ammonia (NH3) when lone pairs are considered?
Tetrahedral
Trigonal planar
Linear
Trigonal pyramidal
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is the bond angle in NH3 slightly less than the theoretical 109.5°?
Due to the absence of lone pairs
Due to the repulsion caused by lone pairs
Because of the presence of triple bonds
Because of the presence of double bonds
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the molecular geometry of CO2?
Tetrahedral
Linear
Trigonal planar
Bent
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How does the presence of lone pairs affect the shape of H2O?
It makes the shape linear
It results in a bent shape
It makes the shape trigonal planar
It results in a tetrahedral shape
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the electron geometry of BH3?
Bent
Linear
Trigonal planar
Tetrahedral
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why does BH3 have a trigonal planar shape?
Due to the presence of triple bonds
Because it lacks lone pairs
Due to the presence of double bonds
Because it has lone pairs
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
