
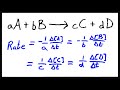
Reaction Rates Quiz
Interactive Video
•
Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Hard

Jennifer Brown
FREE Resource
10 questions
Show all answers
1.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the initial concentration of NH3 in the given problem?
6 M
2 M
8 M
4 M
2.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Which of the following best describes the rate equation for a reaction?
Rate is the sum of concentrations over time.
Rate is the difference in time over concentration.
Rate is the change in concentration over time.
Rate is the product of concentrations over time.
3.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
Why is it important to balance a chemical equation before calculating reaction rates?
To ensure the reaction is spontaneous.
To find the temperature of the reaction.
To make sure the rate equation is applicable.
To determine the color of the reactants.
4.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the balanced coefficient for NH3 in the given reaction?
4
2
1
3
5.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
How is the change in concentration of NH3 calculated?
Initial concentration minus final concentration
Final concentration minus initial concentration
Sum of initial and final concentrations
Product of initial and final concentrations
6.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the time interval given for the reaction?
15 seconds
10 seconds
20 seconds
5 seconds
7.
MULTIPLE CHOICE QUESTION
30 sec • 1 pt
What is the calculated average reaction rate for NH3?
0.01 M/s
0.02 M/s
0.03 M/s
0.04 M/s
Access all questions and much more by creating a free account
Create resources
Host any resource
Get auto-graded reports

Continue with Google

Continue with Email

Continue with Classlink

Continue with Clever
or continue with

Microsoft
%20(1).png)
Apple
Others
Already have an account?
