

Unit 3: Chemical Bonds
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Easy
Standards-aligned
Marcela Diaz
Used 20+ times
FREE Resource
37 Slides • 21 Questions
1
Unit 3: Chemical Bonds
2
Open Ended
9/23/2020
What are the 3 subatomic particles found in an atom? what are the charges of these particles?
3
Why do atoms form bonds?
To complete their outer shell (octet: 8 valence electrons)
To become more stable

4
Multiple Choice
Which family on the periodic table has a complete octet?
Alkali Metals
Alkaline Earth Metals
Transition Metals
Halogens
Noble Gases
5
How are bonds formed?
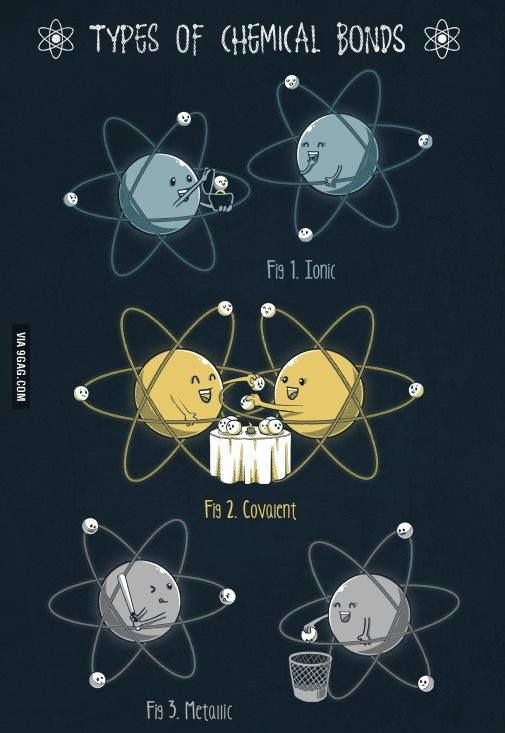
A chemical bond forms when atoms transfer or share e-
Three Types:
ionic, covalent, metallic
6
Ionic Bond
e- are transferred
involves ions (metal and nonmetal elements)

7
Ionic Bond
Metals lose their valence e- and become cations (+)
Nonmetals gain those e- and become anions (-)

8
Ionic Bond
Metals lose their valence e- and become cations (+)
Nonmetals gain those e- and become anions (-)

9
Multiple Choice
Which of the following elements would LOSE electrons?
Na
Cl
F
S
10
Multiple Choice
Which of the following elements would GAIN electrons?
Be
Cl
Li
K
11
NaCl
Na is a metal therefore it's going to LOSE its outer e-
Cl is a nonmetal therefore it's going GAIN the e-

12
NaCl
Na LOST its 1 e- and became a cation (+)
Cl GAINED 1 e- and became an anion (-)

13
Why do atoms become cations when they LOSE e-?
More protons (+) than e- (-)

14
Why do atoms become anions when they GAIN e-?
More e- (-) than protons (+)

15
Metals lose however many valence e- they have
e- in the outermost shell

16
Multiple Choice
How many e- will Sodium lose?
1
2
3
4
5
17
Multiple Choice
How many e- will Aluminum lose?
1
2
3
4
5
18
Multiple Choice
How many e- will Magnesium lose?
1
2
3
4
5
19
Nonmetals GAIN however many valence e- they need to get to 8
Complete their octect (outermost shell)

20
Multiple Choice
How many e- does Fluoride gain?
1
2
3
5
7
21
Multiple Choice
How many e- does Oxygen gain?
1
2
3
5
7
22
Multiple Choice
How many e- does Phosphorus gain?
1
2
3
5
7
23
Complete Ions Table assignment
24
Open Ended
Phosphorus
(valence e-, metal or nonmetal, gain or lose e-, cation/anion, symbol and charge)
25
Lewis Dot Structures
LDS
26
For today you need...
Paper/ notebook
Writing utensil
Periodic Table

27
LDS
show only the valence e- (outer e-)
Begin on the right and go counterclockwise
Each side gets 1 valence e- before doubling up

28
Draw the LDS for the following elements:
1. Helium
2. Fluorine
3. Nitrogen

29
LDS for ionic compounds
metal (+) and nonmetal (-)
30
Multiple Choice
What is happening to the electrons in an ionic bond?
They are being transferred
They are being shared
31
Multiple Choice
In an ionic compound, do metal elements lose or gain e-?
Metals lose e-
Metals gain e-
32
Multiple Choice
When metals LOSE e-, they become...
negative ions called anions
positive ions called cations
negative ions called cations
positive ions called anions
33
Multiple Choice
In an ionic bond, nonmetal elements...
lose e- and become cations
gain e- and become anions
34
LDS for ionic compounds
35
Draw LDS for each atom
Metal element(s) will lose valence-e
Nonmetal element(s) will gain those e- to complete their octet
Redraw LDS showing the transfer of e- and the ion charge
36
Ex 1)
Draw the lewis dot structure for NaCl

37
Ex 2) MgO

38
Ex 3)
Potassium Sulfide (K2S)

39
Open Ended
Exit Ticket: Using the whiteboard app, draw the following LDS:
1) LiF
2) Na2O
3) BaO
40
How are bonds formed?
A chemical bond forms when atoms transfer or share e-
Three Types:
ionic, covalent, metallic
41
Covalent Bonds
e- are SHARED (no ions or charges)
nonmetal elements only

42
Covalent Bond
nonmetal elements ONLY
Ex: CO, H2O

43
Multiple Choice
Which of the following is covalent?
KF
NO2
44
Multiple Choice
Which of the following is covalent?
MgCl2
NH3
45
Multiple Select
Select all of the COVALENT bonds
NaCl
CH4
CO2
Al2O3
SF6
46
LDS for covalent bonds
nonmetals share e-
47
e- sharing is represented using a dash
each dash represents 2 e- being shared

48
Single bond: 2 e- shared
Double Bond: 4 e- shared
Triple bond: 6 e- shared

49
Atoms will share e- in a way that completes their octet (8 valence e-)
*EXCEPTION: H and He, they only need 2 valence e- to be stable!

50
Ex1) Draw the LDS for water
51
Step 1: Draw skeleton structure
The atom that needs the MOST e- goes in the center
*Hydrogen is never the central atom
52
Step 2: Draw each atom's LDS
Pro-tip: use a pencil
53
Step 3: Connect (pair up) electrons
Use a dash to indicate e- being shared
54
Step 4: Check octets
All atom's should have a complete octet (8)
e- being shared count for both elements
*H and He (only need 2)
55
Open Ended
Draw the LDS for O2
56
Metallic Bonds
Metals ONLY
e- are mobile

57
e- move freely through metal
This is how electricity is conducted
e- carry electric current when they move

58
Open Ended
Exit Ticket: Draw the LDS for the following:
CO2
F2
NH3
Unit 3: Chemical Bonds

Show answer
Auto Play
Slide 1 / 58
SLIDE
Similar Resources on Wayground

52 questions
português - Termos da oração
Presentation
•
KG

57 questions
Electron Configuration
Presentation
•
10th Grade

56 questions
Energy Changes in Chemical Reactions
Presentation
•
9th - 10th Grade

56 questions
Enthalpy Change in Chemical Reactions
Presentation
•
9th - 10th Grade

52 questions
Air Distribution Systems
Presentation
•
KG

54 questions
Verbos reflexivos
Presentation
•
10th Grade

51 questions
Reactions of Alcohols 2021-22
Presentation
•
11th Grade

53 questions
G10 Unit 3 RW 2 The Urban Visionary
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade