

Quizizz Lesson: Atomic Structure and the Periodic Table
Presentation
•
Chemistry
•
8th - 12th Grade
•
Easy
Standards-aligned
Ms. McOsker
Used 56+ times
FREE Resource
10 Slides • 8 Questions
1
Quizizz Lesson
Atomic Structure and the Periodic Table

You will need your class notes and your periodic table.
2
The Periodic Table is organized by increasing atomic number.
Atomic Number = Number of Protons
There are 7 rows
There are 18 groups.
Each group has the same number of valence electrons

3
Poll
How many Groups are in the Periodic Table?
2
7
8
18
4
Poll
What is the maximum number of energy levels in an atom?
We have no way to know
up to 7
Each row adds another energy level
up to 18
5
Models: An atomic image focusing on electrons
The nucleus is a single ball in the center
The large gold spheres represent electrons
The rings represent energy levels

6
Multiple Choice

What is the atomic number for this atom?
2
8
7
5
7
Open Ended

What is this element?
8
Model of a Nitrogen Atom
How do you know?
7 total electrons = 7 protons = atomic number 7
OR
5 valence electrons - Group 15
2 rings - second row of the Periodic Table

9
Can you identify these two atoms?

Write your guess on a piece of scrap paper. Then check it on the next slide.
One Valence Electron (Group 1)
Two Rings so second row of periodic table
One Valence Electron (Group 1)
Three Rings so third row of periodic table
10

First atom
Group 1, Second Row = Lithium
It also has 3 total electrons so Atomic Number of 3 = Lithium
Second atom
Group 1, Third Row = Sodium
It also has 11 total electrons so Atomic Number of 11 = Sodium
11
What are these elements? Answer on scrap paper, then check on the next slide.
Top: Valence Electrons = ______, Rings = 2
Element =
Lower: Valence Electrons = ____ Rings = 3
Element =

12
What are these elements?

Top: Valence Electrons = 7,
Rings = 2
ELEMENT = Fluorine
Lower: Valence Electrons = 7
Rings = 3
ELEMENT = Chlorine
13
Nobel Gases have full outer energy levels
The Octet Rule
The first energy level can hold only 2 electrons
All others want 8 to be stable (happy)
An atom want either a full or an empty outer energy level

14
Multiple Choice

TRUE OR FALSE Lithium and Beryllium have the same number of valence electron.
TRUE
FALSE
15
Multiple Choice
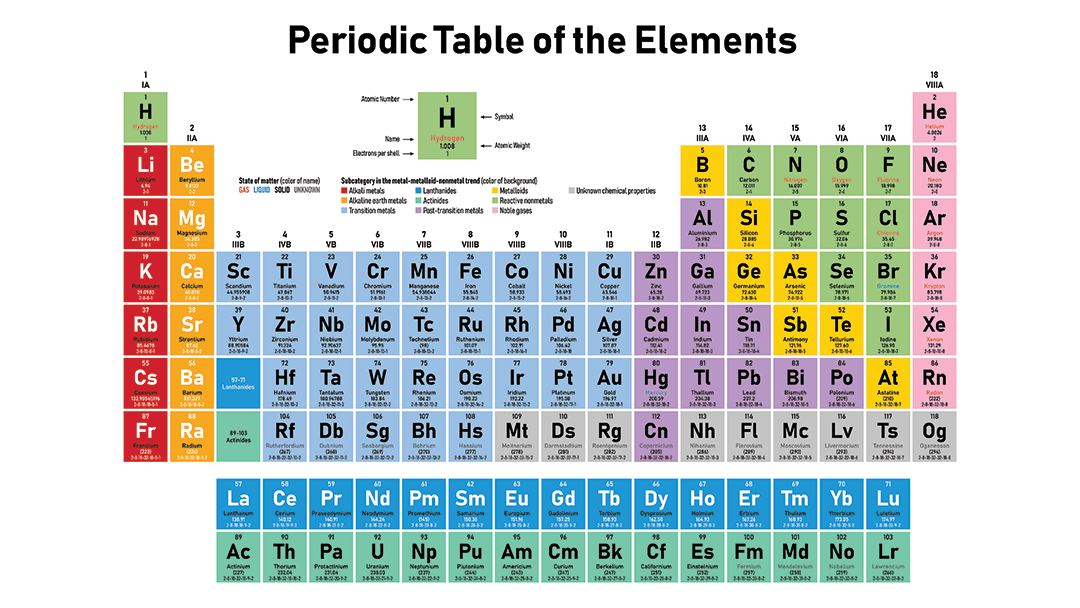
How many valence electrons does Na have?
1
5
6
11
16
Multiple Choice
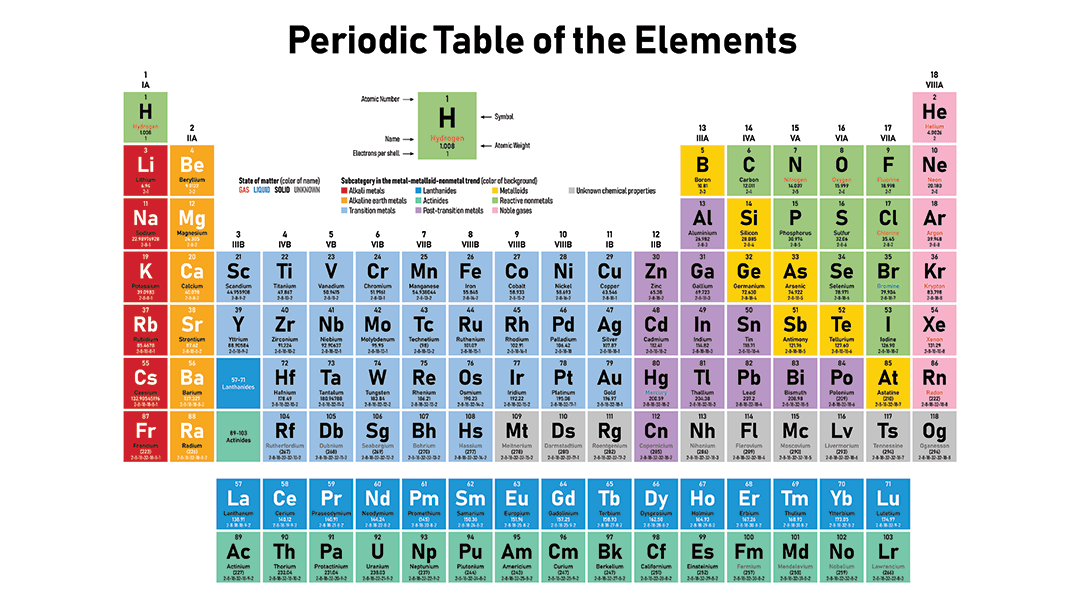
Chlorine ( Cl) has _________ valence electrons.
3
5
7
8
17

Atoms form bonds to be stable. These are now compounds.
18
Multiple Choice
Valence electrons play a key role in chemical reactions their number increases from left to right across a row of the periodic table.
TRUE
FALSE
Quizizz Lesson
Atomic Structure and the Periodic Table

You will need your class notes and your periodic table.
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

13 questions
Ch. 5.3 - Physics and the Quantum Mechanical Model
Presentation
•
9th - 12th Grade

15 questions
Chemistry 1st Six Weeks Intervention_RM
Presentation
•
9th - 12th Grade

13 questions
Quantum Numbers and Electron Configurations
Presentation
•
9th - 12th Grade

12 questions
Mole Conversions Lesson
Presentation
•
9th - 12th Grade

12 questions
Lesson Mole Conversions
Presentation
•
9th - 12th Grade

15 questions
A Evolução dos Modelos Atômicos
Presentation
•
9th - 12th Grade

10 questions
PERIODIC TABLE BASICS
Presentation
•
9th - 12th Grade

19 questions
Chemical Bonding
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade