

Chapter 3 Review
Presentation
•
Science
•
8th Grade
•
Hard
Christiaan Heunis
Used 5+ times
FREE Resource
14 Slides • 19 Questions
1
Chapter 3 Review
- Physical and Chemical Changes
- Chemical Reactions
- Balancing Chemical Equations
- Types of Chemical Reactions
- Endo- and Exothermic Reactions
- Rates of Chemical Reactions

2
Expectations of the lesson
During this lesson you will review the KEY CONCEPTS of Chemical Reactions.
Each KEY CONCEPT will be briefly explained followed by questions to answer.
3
Physical and Chemical Change
A physical change occurs when matter changes its physical properties but not its chemical composition.
A chemical change occurs whenever matter changes into new substances with different chemical properties.

4
Multiple Choice

Can you identify the type of Change?
Physical Change
Chemical Change
5
Multiple Choice
Which picture shows a PHYSICAL change?
6
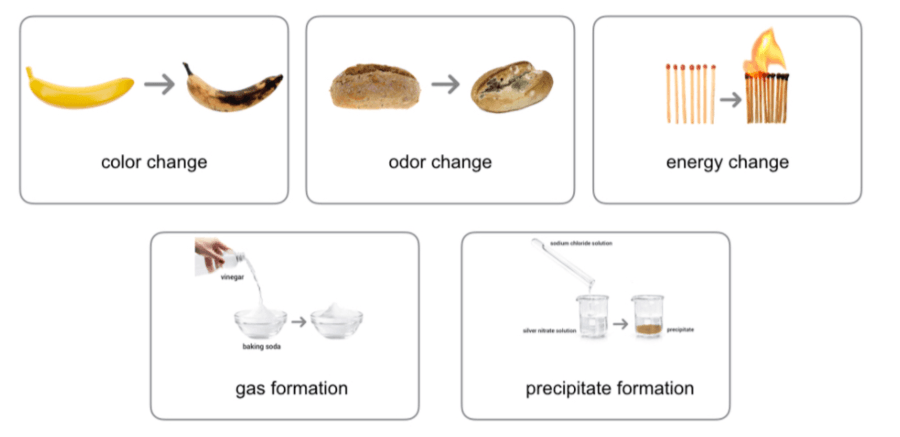
Signs of a Chemical Reaction occurring
7
Multiple Select
Which of the following are signs of CHEMICAL CHANGE?
change in odor
change in shape
change in color
formation of bubbles/gas
8
Fill in the Blanks
Type answer...
9
Chemical Reactions
A chemical reaction is a process by which chemical bonds between atoms are broken and new ones are formed. In chemical reactions, substances change into different substances.
A chemical equation is a statement , used to show changes during a specific chemical reaction.
A chemical equation involves:
- the chemical formula of each substance in the reaction
- an arrow that indicates the process of change, it can be read as "yields" or "reacts to form"

10
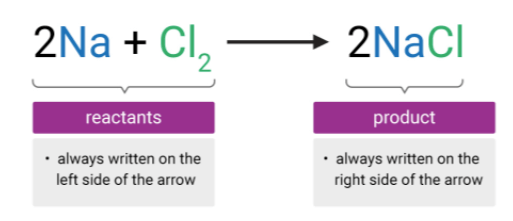
Look at the description of REACTANTS and PRODUCTS
11
Multiple Choice
Identify the reactant in the following chemical equation.
2Mg + O2
2MgO
12
Multiple Choice
Identify the PRODUCTS in the following chemical equation.
Mg + CuSO4
MgSO4 + Cu
13
New Substances
ATOM ARRANGEMENT: The atoms of iron and sulfur are rearranged and new bonds can form to produce iron sulfide.
BOND BREAKING: Each water molecule is made up of two hydrogen atoms joined to one oxygen atom which can decompose by breaking the bonds.
BOND FORMATION: In a compound, the atoms are joined together by strong forces called chemical bonds.

14
Multiple Select
Identify all the ways new substances can be formed.
Atom Rearrangement
Bond Breaking
Bond Formation
15
Multiple Choice
Which statement is true for a chemical reaction?
no new substances are formed
separate stones from sea water
new substances are formed
it is usually easy to reverse the change
16
Multiple Choice
In the reaction below,
CO2 is the ...C + O2 → CO2
electron
bond
reactant
product
17
Fill in the Blanks
Type answer...
18
Balancing Chemical Equations
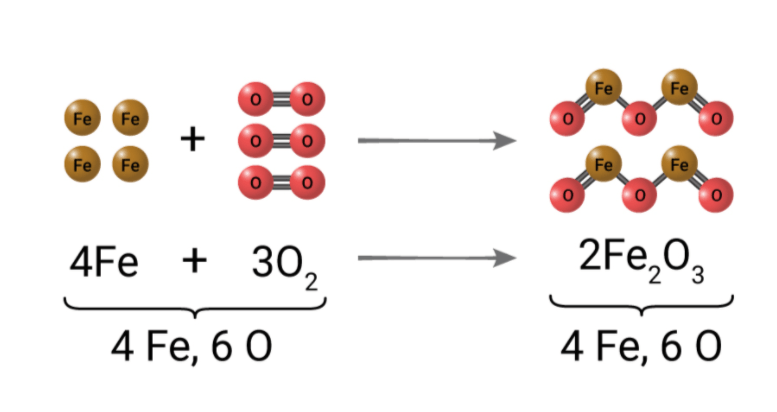
The law of conservation of mass states that mass is never lost or gained in chemical reactions. The total mass of products at the end of the reaction is equal to the total mass of the reactants at the beginning. This is because no atoms are created or destroyed during chemical reactions.

19
How to Balance an equation
In a balanced chemical equation, the total number of atoms remains the same before and after the reaction.
How does a chemical equation for any reaction model that mass is conserved during a chemical reaction?
20
STEP 1: Determine the number of atoms of each element in the reactants and in the products.
STEP 2: Count the number of atoms on each side of the equation.
- If the numbers are equal, then the equation is balanced and if they are not equal, the equation is not balanced.
STEP 3: Add coefficients to the chemical formulas to balance the equation.
STEP 4: Write the balanced chemical equation.

21
Multiple Choice
...... H2+....... O2 → ...... H2O
Balance the following equation.
2H2 + 2O2 → 2H2O
2H2 + O2 → 2H2O
H2+ 2O2 → 2H2O
22
Multiple Choice
...... NH3 → ...... H2+..... N2
Balance the following equation.
2NH3 → 3H2 +N2
2NH3 → H2 +2N2
3NH3 → 3H2 +2N2
23
Multiple Choice
...... CuCO3 → ...... CO2+..... CuO
Balance the following equation.
CuCO3 → 2CO2+2CuO
2CuCO3 → 2CO2+CuO
CuCO3 → CO2+CuO
24
Types of Chemical Reactions
Synthesis Reaction: A synthesis reaction occurs when two elements or compounds combine to form one new compound.
Decomposition Reaction: A decomposition reaction occurs when one reactant breaks down into two or more products.

25
Types of Chemical Reactions
Single-replacement Reaction: In a single replacement reaction, one element replaces another element in a compound.
Double-replacement Reaction: In a double replacement reaction, the two negative ions of two compounds switch places.

26
Types of Chemical Reactions
Combustion Reaction: In a combustion reaction, a substance reacts with oxygen and releases energy in the form of heat and light.

27
Multiple Choice
Which is the correct general equation for a decomposition reaction?
28
Multiple Choice
Identify the type of reaction for the following equation.
2Na + Cl2 → 2NaC
Combustion
Double Replacement
Synthesis
Single Replacement
29
Multiple Choice
Identify the type of reaction for the following equation.
CuSo4+Mg → MgSO4+Cu
Combustion
Double Replacement
Synthesis
Single Replacement
30
Exothermic and Endothermic Reactions
An exothermic reaction is a chemical reaction that releases thermal energy.
An endothermic reaction is a chemical reaction that absorbs thermal energy.

31
Fill in the Blanks

Type answer...
32
Fill in the Blanks

Type answer...
33
Poll
I feel confident in my knowledge of Chemical reactions and Equations
I feel very confident
I feel I need more practice
I do not feel confident
Chapter 3 Review
- Physical and Chemical Changes
- Chemical Reactions
- Balancing Chemical Equations
- Types of Chemical Reactions
- Endo- and Exothermic Reactions
- Rates of Chemical Reactions

Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

25 questions
Gravity & Newton's Laws of Motions
Presentation
•
8th Grade

26 questions
Counting Atoms with Subscripts
Presentation
•
8th Grade

26 questions
Counting Atoms & Chemical Formulas
Presentation
•
8th Grade

25 questions
Physical and Chemical Changes Review
Presentation
•
8th Grade

27 questions
Physical vs Chemical Changes Notes
Presentation
•
8th Grade

23 questions
Rocks and the Rock Cycle
Presentation
•
8th Grade

28 questions
Structures of Matter
Presentation
•
7th - 8th Grade

27 questions
Cell Division
Presentation
•
7th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade