

Density and Relative Density
Presentation
•
Physics
•
10th - 12th Grade
•
Practice Problem
•
Medium
Standards-aligned
r Henderson
Used 79+ times
FREE Resource
16 Slides • 24 Questions
1
Density and Relative Density CXC

2
Observation
Two identical flask are shown one filled with water and the other with kerosene. When measured with an electronic balance the flask filled with water is found heavier than that filled with kerosene. Why?
The answer is in finding the
mass per unit volume of
kerosene and water
in respective flasks.

3
Density; ρ = m/v
Density is referred to as the mass per unit volume. It is a measure of how tightly packed and how heavy the molecules are in an object.

4
Density depends on
mass (usually expressed in grams)
volume (usually expressed in cm3 or liters)
1000cm3 = 1L => 1cm3= 1mL

5
How to find the density
Find the mass of object ; m
Find the volume of object; v
Divide Density;ρ= m/v

6
Guided Practice: 1
A block of ice with volume 5.5m3 has a mass of 5060kg.
Find the density of ice.
Volume of block=5.5m3
Mass of block= 5060 kg
Density = mass /volume
= 5060 kg/5.5m3.
= 920 kg/m3.
The density of ice is 920 kg/m3.
7
Why does ice floats on water?
Pure water has its highest density 1000 kg/m3.
This explains why ice floats on water.

8
Guided Practice:2
A silver cylindrical rod has a length of 0.5m and radius of 0.4m. Find the density of the rod if its mass is 2640kg.
Mass of cylinder =2640 kg
Volume of cylinder = πr²h
= 3.14 x 0.4 2 x 0.5
= 0.2512m3
Density = mass/volume
= 2640 kg/0.2512m3
=10509 kg/m3.
9
Guided Practice:3
A stone has a mass of 112.5g.When the stone is totally immersed in a measuring cylinder containing water the displaced water moves from 50cm3 to 95cm3.Find the density of the stone.
Mass of the stone =112.5g
Volume of stone = 95cm3-50cm3=45cm3
Density = mass/volume
= 112.5g/45cm3
= 2.5g/cm3.
10
Guided Practice:4
Beaker contain 262.5cm3 of a certain liquid weigh 410g,if the mass of an empty dry beaker is 200g. Find the density of the liquid.
Mass of liquid=410g-200g=210g
Volume of liquid=262.5cm3.
Density=mass/volume
=210g/262.5cm3
=0.8g/cm3.
11
Relative Density
the density of a substance compared to that of water
density of substance/density of water
mass of any volume of a substance/ mass of equal volume of water
it is a dimensionless quantity
it can never be zero.
12

13
Multiple Choice
What is mass?
A property of matter that is measured in cm3.
A property of matter that is the amount of atoms and molecules in an object.
A property of matter that describes how tightly packed an object's atoms are.
A property of matter that describes how much space something takes up.
14
Multiple Choice
15
Multiple Choice
What is the first thing you should do when using a triple beam balance?
Clean the object before placing it on the pan.
Place the object on the pan and make sure the pointer and zero are align.
Move the 1's rider to the right.
Zero out the balance using the adjustment knob.
16
Multiple Choice
17
Multiple Choice
A graduated cylinder measures ______________.
mass
matter
water
volume
18
Multiple Choice
19
Multiple Choice

20
Multiple Choice
Density _______________
A property of matter which describes if an object sinks or floats.
A property of matter which is measured on a triple beam balance.
A property of matter which describes how tightly packed an object's molecules are.
A property of matter which is measured with a ruler.
21
Multiple Choice
22
Multiple Choice
SI unit of density is
kgm-4
kgm-3
kgm-2
kgm
23
Multiple Choice
24
Multiple Choice
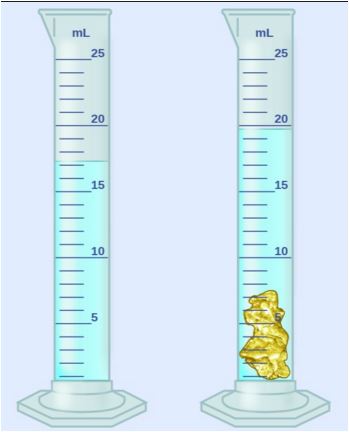
25
Multiple Choice

26
Multiple Choice
Find the volume if a substance has a density of 19.3g/cm3 and a mass of 10g.
.52cm3
193cm3
1.93cm3
29.3cm3
27
Multiple Choice
A material with a density of 5g/cm3 and a volume of 20cm3 has a mass of
25g
4g
50g
100g
28
Multiple Choice
A material with a density of 5g/cm3 and a volume of 20cm3 has a mass of
25g
4g
50g
100g
29
Multiple Choice
30
Multiple Choice
31
Multiple Choice

Which object has the greatest density?
Box with blue particles
Box with red particles
The density of both boxes are equal
32
Multiple Choice
What is the unit for Density?
Grams (g)
Milliliters (mL)
Grams/Milliliter (g/mL)
33
Multiple Choice
The physical property relating the amount of matter in a substance to the volume of the substance.
classify
density
volume
mass
34
Multiple Choice
The comparison of the density of one material as it relates to another; frequently the comparison is to the density of water.
relative density
density
physical property
substance
35
Multiple Choice
A liquid has a density of 626 kg/m3. Work out its relative density.
0.844
0.721
0.586
0.626
36
Relative Density
the density of a substance compared to that of water
density of substance/density of water
37
Relative Density
mass of any volume of a substance/ mass of equal volume of water
38
Relative Density
it is a dimensionless quantity
it can never be zero.
39
Multiple Choice

40
Relative Density
the density of a substance compared to that of water
density of substance/density of water
mass of any volume of a substance/ mass of equal volume of water
it is a dimensionless quantity
it can never be zero.
Density and Relative Density CXC

Show answer
Auto Play
Slide 1 / 40
SLIDE
Similar Resources on Wayground

35 questions
Solubility
Presentation
•
9th - 12th Grade

36 questions
Act Science
Presentation
•
9th - 12th Grade

35 questions
ภาพล้อเลียนและการ์ตูน 1
Presentation
•
9th - 12th Grade

33 questions
Exponential Functions Intro/Review
Presentation
•
9th - 12th Grade

37 questions
Balancing Chemical Equations
Presentation
•
9th - 12th Grade

35 questions
Acids and Bases
Presentation
•
10th - 12th Grade

31 questions
Intro Simple Harmonic Motion
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Physics

50 questions
Honors Physics - 2nd Semester Finals Review - Part 1
Quiz
•
11th Grade

30 questions
Kinetic Energy Question
Quiz
•
11th Grade

50 questions
Honors Physics - 2nd Semester Finals Review - Part 2
Quiz
•
11th Grade

80 questions
2nd Semester Finals Review
Quiz
•
11th Grade

45 questions
Spring 2026 Final Exam
Quiz
•
9th - 12th Grade

17 questions
Newton's Laws
Quiz
•
KG - University