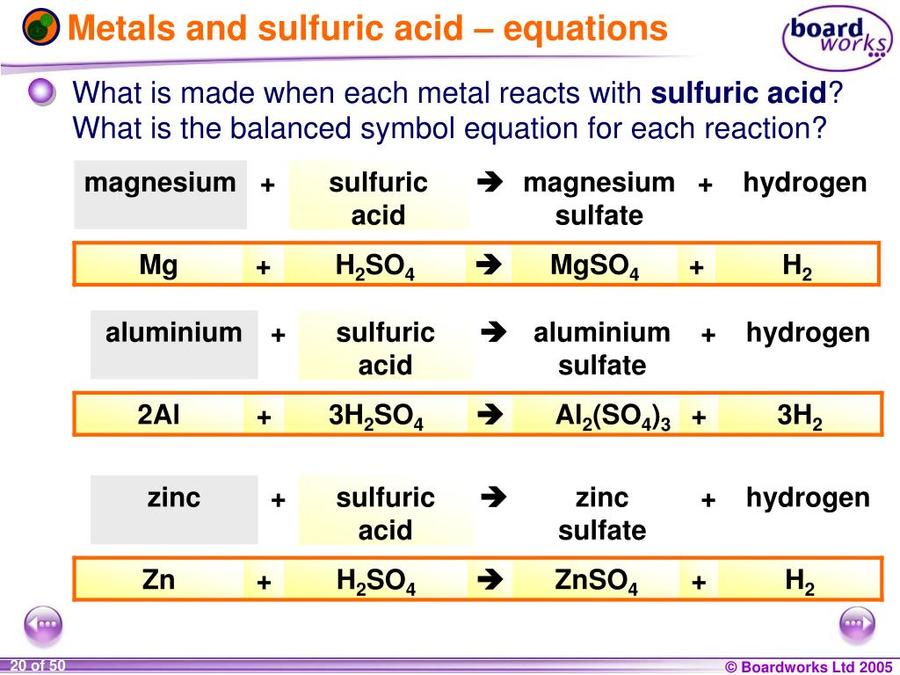
Reacting metal with acid
Presentation
•
Chemistry
•
8th Grade
•
Medium
Michelle Zou
Used 32+ times
FREE Resource
8 Slides • 10 Questions
1
2
3
Fill in the Blanks
4
Fill in the Blanks
5
Fill in the Blanks
6
Fill in the Blanks
7
Fill in the Blanks
8
9
10
11
Fill in the Blanks
12
Fill in the Blanks
13
Fill in the Blanks
14
Fill in the Blanks
15
Fill in the Blanks
16
17
18
Show answer
Auto Play
Slide 1 / 18
SLIDE