

Isotopes
Presentation
•
Chemistry, Science
•
8th - 9th Grade
•
Medium
Standards-aligned
Aaishah V
Used 444+ times
FREE Resource
5 Slides • 6 Questions
1
Isotopes

2
Isotopes
atoms with the SAME number of PROTONS but DIFFERENT number of NEUTRONS
3
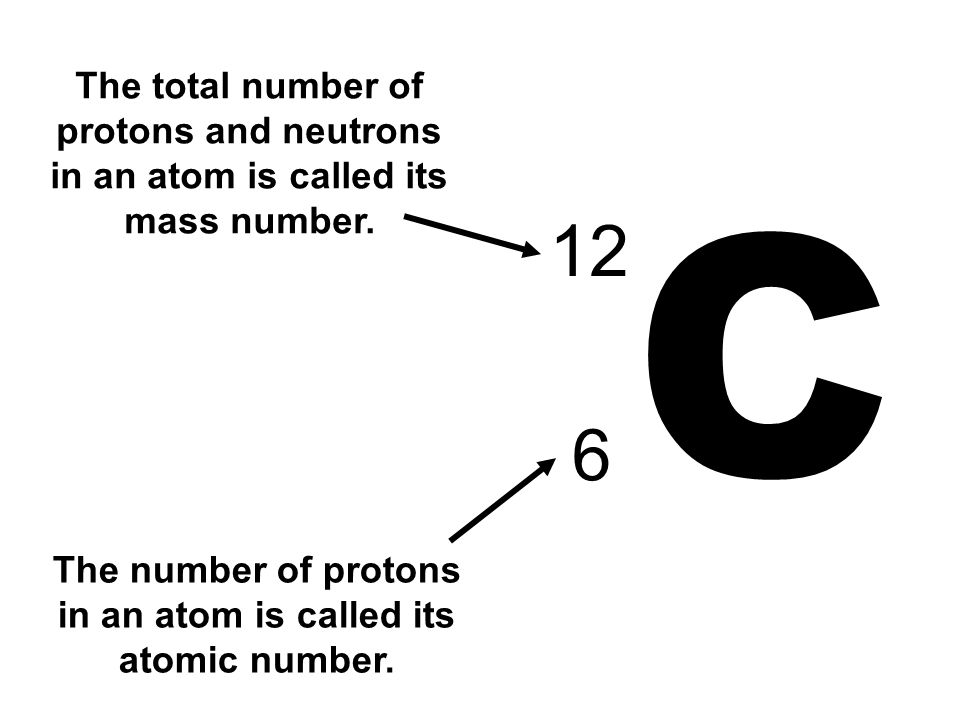
Carbon 12
Consider the Atom Carbon, the atomic number tells us the number of protons.
How many protons in Carbon 12?
4
Multiple Choice
What is the correct definition for isotopes?
atoms with the same number of protons and neutrons
Atoms with the same number of protons but different number of neutrons
Atoms with the same number of neutrons but different number of protons
Atoms with no mass
5
Multiple Choice

The atomic number indicates the number of neutrons
The atomic number indicates the isotope number
The atomic number indicates the number of protons
The atomic number indicates the number of protons and neutrons
6
Multiple Choice

If you want to determine the total number of protons and neutrons in an atom, you can refer to the
atomic mass
atomic number
symbol
charge
7
Lithium isotopes
What do you notice with these 3 isotopes of Lithium?

8
Multiple Choice

What is the difference between these 3 Lithium isotopes?
no difference
they have the same atomic number
they have the same mass number and atomic number
they have different mass numbers
9
Fill in the Blanks
Type answer...
10
Fill in the Blanks
Type answer...
11
The atomic mass of an element is the weighted average of the masses of all its isotopes that occur naturally
Isotopes

Show answer
Auto Play
Slide 1 / 11
SLIDE
Similar Resources on Wayground

10 questions
1.2 Metric Conversions
Presentation
•
8th Grade

11 questions
Moon Phases
Presentation
•
8th Grade

11 questions
Scientific Method
Presentation
•
9th Grade

7 questions
Category 1 Review (8.5B)
Presentation
•
8th Grade

11 questions
Preparing to Balance Equations
Presentation
•
8th Grade

10 questions
Pollution of air and water
Presentation
•
8th - 9th Grade

8 questions
The Chesapeake Bay Resources Review
Presentation
•
8th Grade

9 questions
Formation of Fossil Fuel
Presentation
•
9th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade

20 questions
Acids and Bases
Quiz
•
8th Grade

20 questions
Le Chatelier's Principle
Quiz
•
9th - 12th Grade

20 questions
Chemical Reactions
Quiz
•
8th Grade

6 questions
DOL: Endothermic and Exothermic Reactions
Quiz
•
9th - 11th Grade

12 questions
Rate of reaction
Quiz
•
8th - 9th Grade