

OBJ 4.7-4.9 Valence Electrons & Dot Diagrams
Presentation
•
Chemistry
•
8th - 10th Grade
•
Easy
Standards-aligned
Kristen Kush
Used 42+ times
FREE Resource
14 Slides • 12 Questions
1
OBJ 4.7-4.9 Valence Electrons & Dot Diagrams
If you answer all the Qs and earn at least a 70% then you'll get a full 10/10. It's better to guess than skip
2
OBJ 4.7: Explain where valence electrons are located within an atom
The electrons in the outermost energy level are most important for bonding and a chemical reactions
These electrons are called valence electrons.
3
Poll
What are valence electrons?
Electrons in the outermost energy level
Responsible for chemical reactions and bonding
Electrons in the inside the nucleus
Not important in chemistry
4
What are valence electrons?
Valence electrons: the electrons located in the outermost energy level of an atom
Responsible for the chemical reactions between elements that form molecules.
5
Multiple Choice
Where are valence electrons?
In the nucleus
In the last energy level
In the first energy level
Not in an atom
6
Using Bohr Notation
The last number represents the number of valence electrons
An atom can have 1-8 Valence electrons (for now)
Example: Nitrogen 2-5
How many valence elctrons?
5!

7
Multiple Choice
Given sodium (Na) Bohr: 2-8-1
How many valence electrons does sodium have?
2
8
1
11
8
Multiple Choice
Given chlorine (Cl) Bohr: 2-8-7
How many valence electrons does chlorine have?
2
8
7
17
9
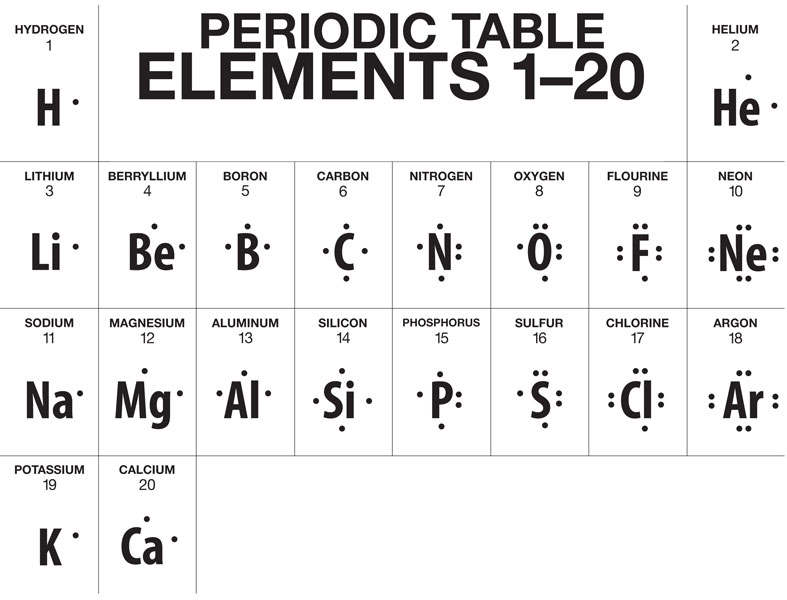
OBJ 4.8: Identify the number of valence electrons in a neutral atom using the periodic table
As we know, the periodic table has patterns
We can use these patterns and the "A" group numbers to determine how many valence electrons are in a neutral element
10

11
Open Ended

What pattern did you notice?
12
Recognizing Patterns
How do we determine the number of valence electrons in a neutral atom?
For all elements located in the “A” groups:
The number in front of the A is equal to the number of valence electrons.
the group “A” number = # of valence e-

13
Multiple Choice
Beryllium (Be) is in group 2A. How many valence electrons does it have?
1
2
4
A
14
Multiple Choice
Krypton (Kr) is in group 8A. How many valence electrons does it have?
2
8
36
18
15
OBJ 4.9: Draw Lewis dot structures for atoms
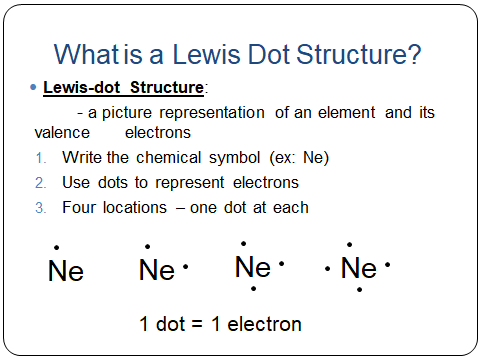
What is a Lewis Dot Structure / Electron Dot Diagram?
a picture representation of an element and its valence electrons
Each dot represents one valence electron
16
17

18
Multiple Choice
Nitrogen is in group 5A so it has 5 valence elctrons. How many dots will be needed for the Lewis Dot Structure?
5
7
0
1
19
Nitrogen
5 valence electrons = 5 dots
4 locations for electrons
Fill the "empty" space first, then pair up after

20
Multiple Choice
Sulfur is in group 6A. Which is the correct Lewis Dot Structure?
21

22
Helium: the "exception"
How many total electrons does helium have?

23
Open Ended
Helium has an atomic number of 2. It is located in group 8A. How many valence electrons do you think it has?
24
Helium
Helium has 2 electrons. So it only has 2 valence electrons.
Elements in group 8A have full orbitals / shells
All other elements in group 8A are said to have an "octet" 8 electrons
Think: 2s2 + 2p6 = 8 electrons
There is only one orbital in the first energy level (1s)
Helium is called a "duet"

25
Poll
Goals of today. Check off the box(es) that apply.
I can explain what/where valence electrons are
I can use the periodic table to identify the number of valence electrons in an element
I can draw Lewis Structures for an element
I cannot do any of the above
26
Open Ended
What question(s) do you have?
OBJ 4.7-4.9 Valence Electrons & Dot Diagrams
If you answer all the Qs and earn at least a 70% then you'll get a full 10/10. It's better to guess than skip

Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

20 questions
Revising and Editing Grammar Review
Presentation
•
8th - 10th Grade

22 questions
Japanese-American Internment Camps
Presentation
•
8th - 10th Grade

20 questions
Classifying Triangles by Angles & Sides
Presentation
•
8th - 10th Grade

20 questions
Photosynthesis
Presentation
•
9th - 10th Grade

20 questions
Scatter Plots
Presentation
•
8th - 9th Grade

18 questions
A1 4-4 Linear Inequalities in Two Variables
Presentation
•
8th - 10th Grade

21 questions
Spanish 2 DOP lesson
Presentation
•
9th - 10th Grade

20 questions
Lesson: Basic States of Matter
Presentation
•
8th - 9th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade