

Chemical Formulas
Presentation
•
Science
•
8th Grade
•
Medium
Standards-aligned
Amber Gilbert
Used 3+ times
FREE Resource
13 Slides • 13 Questions
1
Chemical Formulas
How are chemical formulas used to identify substances?

2
ELEMENTS
A chemical that cannot be broken down into another substance.
Each unique capital letter represents a different element.

3
ELEMENTS
How many elements are in glucose?

4
ATOMS
The smallest part of a chemical element.

5
MOLECULES
Combining atoms creates molecules.

6

7
Formula for Molecules
An expression which states the number and type of atoms present in a molecule of a substance.
*H2 tells you there are 2 atoms of hydrogen.

8
H2O tells you
2 atoms of hydrogen
and
1 atom of oxygen.

9
SUBSCRIPTS
The small numbers after the element’s name.
No subscript means there is only 1 atom present. (The one is not needed.)

10
Counting Atoms
1. List the elements.
Cu
S
O
2. Record the atoms of each element.
Cu-1
S-1
O-4

11
Multiple Choice
Which of the following chemical formulas contains exactly 4 hydrogen atoms?
CH3COONa
NH4OH
CH3CH2Cl
CH3CHO
12
Multiple Choice
How many atoms of carbon are in the formula CH3CH2CH2OH?
3
1
7
4
13
Multiple Choice
How many atoms of hydrogen are in the formula CH3CH2Br?
5
3
2
7
14
Multiple Choice

Which elements are in the formula shown above?
Tin and Chlorine
Tin, Carbon and Lithium
Sulfur, Nitrogen and Chlorine
Sulfur, Nitrogen, Carbon and Lithium
15
Multiple Choice

Which of the following accurately describes the chemical formula of the molecule shown above?
3CH8O
C3H7O
C4H8O2
C3H8O
16
Multiple Choice
17
Multiple Choice
Sodium sulfate is used in laundry detergent. It is a compound that contains 2 atoms of sodium, 1 atom of sulfur, and 4 atoms of oxygen. What is the correct chemical formula for sodium sulfate?
NA2SO4
Na2S2O4
Na2SO4
Na2SO8
18
Multiple Choice

19
Multiple Choice
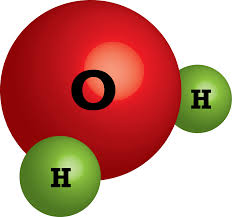
20
Multiple Choice

21
Multiple Choice
What elements are represented in this chemical formula? Fe2O3
Iron, Oxygen
Francium, Europium, Oxygen
Ferrium, Oxygen
Berylium, Oxygen
22
Multiple Choice
How many atoms of Carbon are in this formula? C6 H12O6
6
1
12
23
Multiple Choice
What does the subscript in a chemical formula tell you?
How many molecules are present
How many atoms are present
How many elements are present
How many neutrons are present
24
Subscripts + Coefficients
Big number at the beginning of this formula is a coefficient.
Represents the number of molecules for that compound

25
Subscripts + Coefficients
This formula means there are 2 H2SO4 molecules.
We know in each molecule there are:
2 hydrogens
1 sulfur
4 oxygens

26
To find the total number of atoms:
multiply each of these atoms by 2.
2 hydrogens x2
1 sulfur x2
4 oxygens x2
Total atoms for this formula = 14

Chemical Formulas
How are chemical formulas used to identify substances?

Show answer
Auto Play
Slide 1 / 26
SLIDE
Similar Resources on Wayground

18 questions
Rocks and Rock Cycle
Presentation
•
8th Grade

18 questions
Cellular respiration vs photosynthesis
Presentation
•
8th Grade

21 questions
Solar System Vocabulary
Presentation
•
8th Grade

21 questions
Net Force Review
Presentation
•
8th Grade

19 questions
Element's Identity & Valence Electrons
Presentation
•
8th Grade

19 questions
Light Interactions with Matter
Presentation
•
8th Grade

19 questions
Taxonomy
Presentation
•
8th Grade

21 questions
Specific heat Capacity
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

10 questions
Exploring the Layers of the Earth
Interactive video
•
6th - 10th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade

20 questions
Cell Organelles and Functions
Quiz
•
6th - 8th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade