

Ch. 11.1 - Describing Chemical Reactions
Presentation
•
Chemistry, Science
•
9th - 11th Grade
•
Medium
Jeremy Bosquez
Used 7+ times
FREE Resource
2 Slides • 12 Questions
1
Ch. 11.1
Describing Chemical Reactions
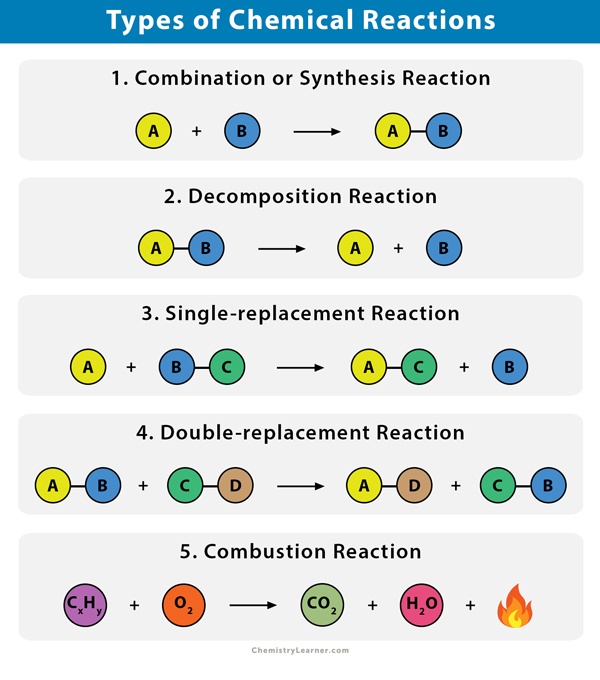
2
3
Fill in the Blank
Type answer...
4
Fill in the Blank
Type answer...
5
Multiple Choice
Sulfur burns in oxygen to form sulfur dioxide. Select the correct skeleton equation for this chemical reaction.
S + O2 --> SO2
S + O --> SO2
S8 + O --> SO2
S2 + O2 --> SO2
6
Multiple Choice
Which of the following is a skeleton equation?
2H2 + CO2 🡪 CH3OH
2H2 + CO 🡪 CH3OH
H2 + CO 🡪 CH3OH
Hydrogen + carbon monoxide 🡪 methanol
7
Multiple Choice
Select the correct skeleton equation for this word equation.
Hydrogen + Sulfur --> Hydrogen Sulfide
H + S --> HS
H + S --> H2S
H2 + S --> HS2
H2 + S --> H2S
8
Multiple Choice
Select the correct coefficients to balance this equation.
_H2 + _S --> _H2S
Hydrogen + Sulfur --> Hydrogen Sulfide
1, 2, 1
1, 1, 1
2, 1, 2
2, 1, 1
9
Multiple Choice
Select the correct skeleton equation for this word equation.
Iron (III) chloride + Calcium hydroxide --> Iron (III) hydroxide + Calcium chloride
FeCl + CaOH --> FeOH + CaCl
FeCl3 + Ca(OH)2 --> Fe(OH)3 + CaCl2
Fe3Cl + Ca2OH --> Fe3OH + Ca2Cl
FeCl3 + CaOH --> Fe(OH)3 + CaCl
10
Multiple Choice
Select the correct skeleton equation for this word equation.
Calcium hydroxide + sulfuric acid --> calcium sulfate + water
CaOH + HSO4 --> CaSO4 + H2O
Ca(OH)2 + H2S --> CaS + H2O
Ca(OH)2 + H2SO4 --> CaSO4 + H2O
Ca(OH)2 + H2SO3 --> CaSO3 + H2O
11
Multiple Choice
Select the correct coefficients to balance the skeleton equation.
_Ca(OH)2 + _H2SO4 --> _CaSO4 + _H2O
1, 1, 1, 2
1, 1, 1, 1
1, 2, 2, 1
2, 1, 2, 1
12
Multiple Choice
What coefficient for H2SO4 is required to balance the following equation?
Ca3(PO4)2 + ____ H2SO4 → 3CaSO4 + 2H3PO4
1
2
3
4
13
Open Ended
Write a balance chemical equation for the following reaction.
Aluminum reacts with aqueous hydrochloric acid to form hydrogen gas and aqueous aluminum chloride.
14
Open Ended
Write a balance chemical equation for the following reaction.
Acetylene gas (C2H2) burns in a welding torch with oxygen to form carbon dioxide gas and water vapor.
Ch. 11.1
Describing Chemical Reactions

Show answer
Auto Play
Slide 1 / 14
SLIDE
Similar Resources on Wayground

11 questions
Ionic Compounds & Covalent Molecules - 10/1
Presentation
•
9th - 11th Grade

10 questions
Absolute Value Equations
Presentation
•
9th - 12th Grade

12 questions
Electrolytes and Non Electrolytes
Presentation
•
10th Grade

10 questions
Lección Ciencias Naturales
Presentation
•
10th Grade

10 questions
Factoring by Grouping
Presentation
•
9th - 11th Grade

11 questions
P-T Diagram Group Practice
Presentation
•
9th - 12th Grade

11 questions
Pythagorean Theorem
Presentation
•
9th - 11th Grade

10 questions
SÂN CHƠI TẾT YÊU THƯƠNG
Presentation
•
KG
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

25 questions
Solutions Test Review
Quiz
•
10th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

15 questions
STAAR Review Quizziz 1
Quiz
•
10th Grade

15 questions
STAAR Wayground #2
Quiz
•
10th Grade

15 questions
Ionic Compounds: Naming and Formula Writing
Quiz
•
9th - 12th Grade

5 questions
DOL IP: Solubility and Precipitation Reactions
Quiz
•
9th Grade

10 questions
Exploring Factors Influencing Solubility
Interactive video
•
6th - 10th Grade