

Ideal Gases And Kinetic Theory U1 M2
Presentation
•
Physics
•
11th Grade
•
Medium
PIERRE ROCK
Used 13+ times
FREE Resource
15 Slides • 15 Questions
1
Ideal Gases And The Kinetic Theory U1 M3

2

3

4

5

6

7

8

9

10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
What is the formula Charles' Law?
V = T
VT = VT
T1 / V1 = T2 / V2
V1 / T1 = V2 / T2
13
Multiple Choice
What remains constant according to Charles’ Law?
Length
Pressure
Temperature
Volume
14
Multiple Choice
In CHARLES'S LAW, volume and temperature have a ______ proportionality.
direct
inverse
linear
exponential
15
Multiple Choice
In CHARLES' LAW, at constant pressure, what happens to temperature of a gas when the volume doubles?
The temperature also doubles.
The temperature decreases to half of its original volume.
The temperature doesn't change.
The temperature quadruples.
16
Multiple Choice
17
Multiple Choice
What is the formula for Boyle's Law?
P1V1=P2V2
P1V1/P2V2
P1V2=P2V1
P1/V1=P2/V2
18
Multiple Choice
In BOYLE'S LAW, when Pressure increases then the Volume must...
Increase
Decrease
19
Multiple Choice
In BOYLE'S LAW, when Volume increases then Pressure must...
Increase
Decrease
20
Multiple Choice
In BOYLE'S LAW, what type of relationship do pressure and volume have?
Direct
Inverse
No relationship
21
Multiple Choice
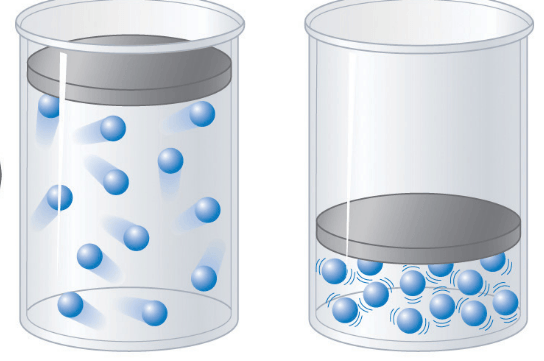
Which container will have a lower pressure?
left
right
they both have the same pressure
22

23

24

25

26

27

28
Multiple Select
Kinetic Theory Equations
Kinetic Theory is used to explain all the gas laws by observing the movement of the particles. Several assumptions are made to derive these equations:
Please tick the correct assumptions below.
(Hint: Use the Note entitled "4 Physics U1 M3 THE KINETIC THEORY OF GASES")
The molecules of the gas can be assumed to be point molecules. This means that they have negligible volume.
There is no intermolecular forces of attraction. The particles are in random motion and the average speed of the random motions produce a constant force (constant
force → constant pressure)
Collisions are (perfectly) elastic, the particles rebound after collision and kinetic energy is conserved.
Impact time is minimum. The rate of change of momentum during the impact gives an average force provided that the time of impact is much less than the time
between impacts.
All molecules of a particular gas are identical.
29
Multiple Select
Kinetic Theory Equations (Continued)
Kinetic Theory is used to explain all the gas laws by observing the movement of the particles. Several assumptions are made to derive these equations:
Please tick the correct assumptions below. (Hint: Use the Note entitled "4 Physics U1 M3 THE KINETIC THEORY OF GASES")
Newton’s Three Laws are adhered to.
There is a sufficiently large number of molecules for statistics to be meaningfully
applied.
30
Poll
How did you find this lesson to be?
This lesson was very informative, please do more like this.
This lesson was very informative, but please do less like this.
This lesson needs more explanations, but I like the format.
This lesson needs more explanations and I do not like the format.
Ideal Gases And The Kinetic Theory U1 M3

Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

22 questions
Tiro Parabólico
Presentation
•
11th Grade

25 questions
Properties of Waves
Presentation
•
10th Grade

21 questions
Electric Field
Presentation
•
12th Grade

24 questions
2-13 Solving Systems of Linear Inequalities
Presentation
•
11th Grade

23 questions
Monday 5/13 Check In
Presentation
•
11th Grade

21 questions
Best Method: Solving Systems of Equations
Presentation
•
7th - 10th Grade

21 questions
Cold War Intro
Presentation
•
KG

20 questions
Work and Power
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

36 questions
6th Grade Math STAAR Review
Quiz
•
6th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade