

The Periodic Table
Presentation
•
Science, Chemistry
•
8th Grade
•
Medium
+1
Standards-aligned
Ginger Sudol
Used 7+ times
FREE Resource
9 Slides • 21 Questions
1
The Periodic Table

2
Learning Objectives: I can...
Identify different atoms by the number of protons in the nucleus
Demonstrate that the number of electrons equals the number of protons in a neutral atom
Explain the meaning of atomic number and atomic mass
3
History of the Periodic Table
In 1869, Russian chemist Dimitri Mendeleev started arranging chemical elements by atomic mass.
Predicted the discovery of other elements and left spaces in the periodic table for them

4
Multiple Choice
How was Mendeleev's periodic table arranged?
by increasing atomic mass
by decreasing atomic mass
by increasing atomic number
by decreasing atomic number
5
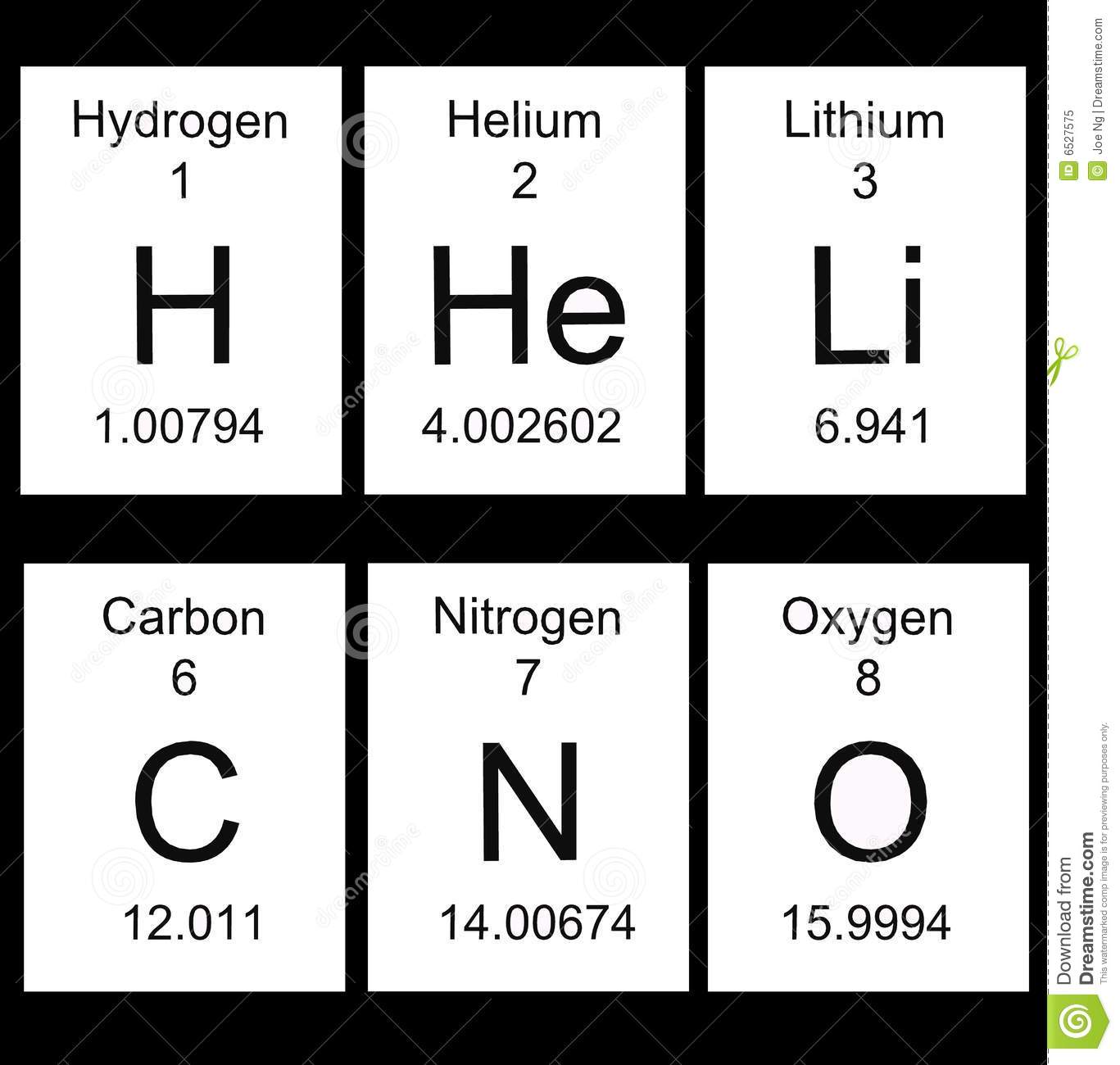
The Periodic Table: First 20 Elements
Many common substances are composed of atoms from these first 20 elements.
Each box contains information about a different element
Shows all the atoms that everything in the known universe is made from

6
An Example: Carbon
Atomic Number: Number of protons in the nucleus of the atom
Atomic mass: the average mass of atoms in an element
Element Name: Usually from a Greek or Latin word for the element or a substance containing the element.
Symbol: Short-hand abbreviation for the element name (starts with a capital letter)

7
Multiple Choice

What is the atomic mass of
iron (Fe)?
26
13
55.845
23.423
8
Multiple Choice

What is the atomic number of Sulfur?
16
2
32.065
86
9
Atomic Mass
Based on the mass of the protons, neutrons and electrons of that element.
Mass of the proton and neutron are about the same, but the mass of the electron is much smaller
Majority of the atomic mass is contributed by the protons and neutrons

10
Atomic Mass
For any element in the periodic table, the number of electrons in an atom always equals the number of protons in the nucleus
Not true for neutrons
Atoms of the same element can have different numbers of neutrons than protons.
They are called isotopes.
Atomic mass is the average of the atomic mass of the isotopes of an element.

11
Carbon
Vast majority of carbon atoms have 6 protons and 6 neutrons
A small percentage have 6 protons and 7 neutrons
An even smaller percentage have 6 protons and 8 neutrons
Since the majority of carbon atoms have a mass very close to 12, the average atomic mass is slightly greater than 12.

12
Multiple Choice
Isotopes of an element have a different number of...
Protons
Neutrons
Electrons
Mass
13
Subatomic Particles
Proton: positively charged particle; located in the nucleus; is the atomic number
Electron: negatively charged particle surrounding the nucleus; equal to the number of protons
Neutron: No charge; has almost the same mass as a proton; equal to or slightly greater than the number of protons
14
Multiple Choice

How many protons are in Sodium?
11
22
12
33
15
Multiple Choice

16
Multiple Choice

17
Multiple Choice

18
Multiple Choice

19
Multiple Choice

What is the atomic mass of Neon?
10
20.18
10.18
9.82
20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice

24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
27
Multiple Choice
28
Multiple Choice

29
Multiple Choice
What is at the center of every atom?
An electron
A compound
A molecule
A nucleus
30
Multiple Choice
The Periodic Table

Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

24 questions
The Nature of Science
Presentation
•
8th Grade

25 questions
Water Quality EOG Review
Presentation
•
8th Grade

21 questions
Seasons
Presentation
•
8th Grade

21 questions
SSA Review
Presentation
•
8th Grade

23 questions
Continental Drift
Presentation
•
8th Grade

23 questions
Properties of Stars
Presentation
•
8th Grade

24 questions
Forces that Shape the Earth
Presentation
•
8th Grade

24 questions
Continental Drift and Plate Tectonics Lesson
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade

15 questions
Symbiotic Relationships
Quiz
•
6th - 8th Grade

10 questions
Food Chain and Food Web
Quiz
•
8th Grade

24 questions
8th grade Science STAAR Review
Quiz
•
8th Grade