

stoichiometry
Presentation
•
Chemistry
•
10th Grade
•
Practice Problem
•
Medium
Standards-aligned
maysaa el harakeh
Used 242+ times
FREE Resource
1 Slide • 15 Questions
1
stoichiometry

2
Multiple Choice
If 55.6 grams of calcium carbonate react in excess sodium hydroxide, how many grams of Na2CO3 are made?
Reaction: CaCO3 + NaOH → Ca(OH)2 + Na2CO3
147.34 gram
52.45 gram
55.9 gram
194.24 gram
3
Multiple Choice
I use mole ratio (coefficients) from balanced chemical equation?
True
False
4
Multiple Choice
I use molar mass when the problem gives me grams?
False
True
5
Multiple Choice
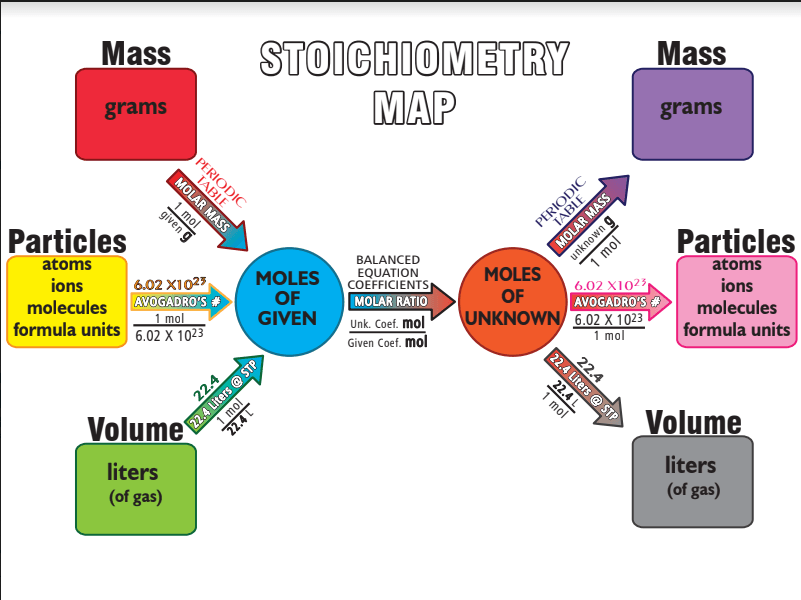
How many step will it take for me to go from grams to moles unknown
1
2
3
4
6
Multiple Choice
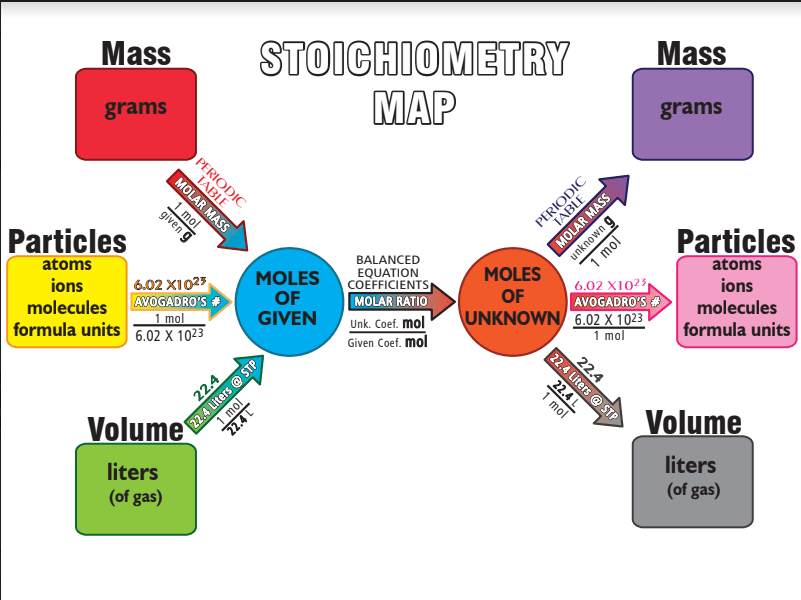
How many steps will it take me to go from grams to grams?
4
3
2
1
7
Multiple Choice

How many step will it take for me to go from moles given to moles unknown?
1
2
3
4
8
Multiple Choice
In the equation 2Al2O3 → 4Al + 3O2, what is the mole ratio of aluminum to oxygen?
10:6
3:4
4:3
2:3
9
Multiple Choice
___KNO3 → ___KNO2 + ___O2
What coefficients are needed to balance the reaction?
2, 2, 1
2, 3, 2
1, 2, 2
1, 3, 1
10
Multiple Choice
Given the equation:
2NaClO3 (s) → 2NaCl (s) + 3O2 (g)
When 2.00 moles of NaClO3 react, how many grams of O2 is produced?
56 g of O2
96 g of O2
64 g O2
32 g O2
11
Multiple Choice
Mg + 2HCl --> MgCl2 + H2
12
Multiple Choice
13
Multiple Choice
How many moles of carbon dioxide are produced from the combustion of 110 g of CH4?
14
Multiple Choice
How many grams of potassium chloride can be produced from 356 g of chlorine and 356 g of potassium bromide?
15
Multiple Choice
I need to add 8 moles of Carbon to the reaction. How many grams of Carbon should I weigh?
16
Multiple Choice
How many grams of hydrogen are produced if 120 g of Na are available?
stoichiometry

Show answer
Auto Play
Slide 1 / 16
SLIDE
Similar Resources on Wayground

11 questions
HAJI, ZAKAT , DAN WAKAF
Presentation
•
10th Grade

11 questions
PSAT Orientation
Presentation
•
10th Grade

10 questions
Percent comp, empirical, molecular formula
Presentation
•
10th Grade

12 questions
Tangent and Secant Segment
Presentation
•
10th Grade

12 questions
Way Maker
Presentation
•
KG

11 questions
Stoichiometry mol to mol
Presentation
•
10th Grade

10 questions
KONSEP MOL
Presentation
•
10th Grade

13 questions
Lewis dot diagram (for elements)
Presentation
•
10th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade