

Introduction to Acids and Bases
Presentation
•
Chemistry
•
10th Grade - University
•
Medium
Jessica Iseman
Used 5+ times
FREE Resource
10 Slides • 28 Questions
1
Introduction to Acids and Bases

2

3
Multiple Select

Which of the following is a property of acids
taste sour
taste bitter
react with metals
turn litmus paper red
turn litmus paper blue
4
Multiple Select
Which of the following are examples of acids?
lemons
soda
baking soda
salt
5

6
Multiple Select
Which of the following are properties of bases?
taste bitter
feel slippery
taste sour
are posionous
turn litmus paper blue
7
Multiple Select
Which of the following are examples of bases?
Fruit
Sour Patch Candy
Baking Soda
Soap
8
Multiple Choice
Do acids and bases conduct electricity?
Only acids conduct electricity
Only bases conduct electricity
Bases and acids conduct electricity
Neither conduct electricity
9

10
Multiple Select
What are the three types of acids and bases
Bronsted-Lowry
Arrhenius
Amphiprotic
Lewis
Polyprotic
11

12
Multiple Choice
When using the Arrhenius model, what types of ions indicate that a compound is a base?
H+1
OH-1
13
Multiple Choice
When using the Arrhenius model, what types of ions indicate that a compound is an acid?
H+
OH-1
14
Multiple Choice
Is the following compound an acid or a base?
H3PO4
acid
base
15
Multiple Choice
Is the following compound an acid or a base?
HI
acid
base
16
Multiple Choice
Is the following compound an acid or a base?
NH4OH
acid
base
17
Multiple Choice
Is the following compound an acid or a base?
HCl
Acid
Base
18

19

20
Multiple Choice
When using the Bronsted Lowry definition of acids and bases, the base will ______________ a hydrogen proton.
donate
accept
21
Multiple Choice
When using the Bronsted Lowry definition of acids and bases, the acid will ______________ a hydrogen proton.
donate
accept
22
Multiple Choice
In the following chemical equation, which compound is the base?
HCl + NH3 → NH4+ + Cl−
HCl
NH3
NH4+
Cl−
23
Multiple Choice
In the following chemical equation, which compound is the acid?
HCl + NH3 → NH4+ + Cl−
HCl
NH3
NH4+
Cl−
24

25
Multiple Choice
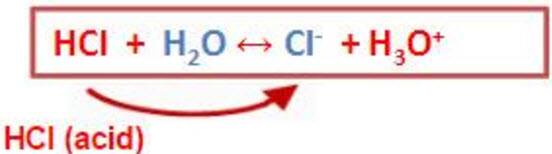
26
Multiple Choice
Which of the following correctly identifies an Acid-Base Pair?
HH4+ , OH-
H2O , OH-
CaCO3 ,CaCl2
HNO3 , H2SO4
27
Multiple Choice
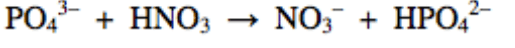
28
Multiple Choice
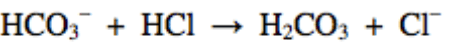
What is the conjugate base in the following reaction?
HCO3-
HCl
H2CO3
Cl-
29
Multiple Choice
What is being donated/accepted between conjugate acid-base pairs?
a hydrogen (H+) ion
a hydroxide (OH-) ion
water
a neutron
30

31
Multiple Select

Classify this acid.
Arrhenius Acid
Polyprotic acid
Lewis Acid
Monoprotic acid
32
Amphiprotic substances
Substances that can act as either an acid or a base.
Also known as amphoteric

33
Multiple Choice
34
Multiple Choice
35
Multiple Choice
NaOH is:
an Arrhenius base
an Arrhenius acid
neither an acid nor a base
both an acid and a base
36
Multiple Choice
CH3NH2 + H2O --> CH3NH3+ + OH-
CH3NH2 can be classified as which of the following? (HINT...follow the H+!!!)
37
Multiple Choice
38
Multiple Choice
Introduction to Acids and Bases

Show answer
Auto Play
Slide 1 / 38
SLIDE
Similar Resources on Wayground

37 questions
The Mole Concept - An Introduction
Lesson
•
9th - 12th Grade

31 questions
14.2 Urinary System
Lesson
•
11th - 12th Grade

35 questions
Add it to water?
Lesson
•
9th - 12th Grade

34 questions
Collision Theory & Reaction Rate Experiment
Lesson
•
KG

33 questions
Periodic
Lesson
•
10th - 12th Grade

34 questions
Macromolecules
Lesson
•
9th - 12th Grade

27 questions
Reaction Rates
Lesson
•
9th - 12th Grade

32 questions
review present simple vs present continuous
Lesson
•
10th Grade - University
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

15 questions
Hargrett House Quiz: Community & Service
Quiz
•
5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Element compound mixture review quiz
Quiz
•
10th - 12th Grade

10 questions
Determining Protons, Neutrons, and Electrons from the Periodic Table
Interactive video
•
6th - 10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
U4 Reaction Types
Quiz
•
10th - 12th Grade

25 questions
Potential Energy Diagram Checkpoint
Quiz
•
11th Grade

15 questions
IP PreAP: Solubility Rules and Precipitation Reactions
Quiz
•
University

46 questions
Quantum, E Config, Periodic Trends
Quiz
•
11th Grade