

Gas Laws: Part 2
Presentation
•
Chemistry
•
10th Grade
•
Easy
ANDREA MEYER MUNOZ
Used 56+ times
FREE Resource
0 Slides • 15 Questions
1
Multiple Choice
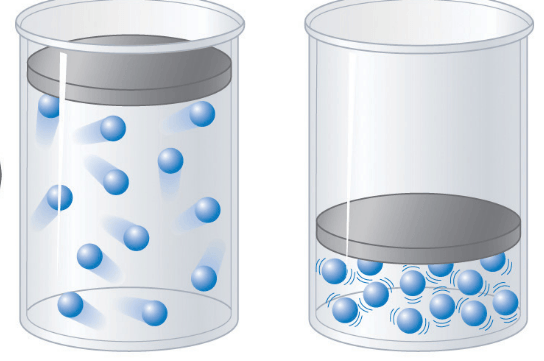
left
right
they both have the same pressure
I don't know
2
Multiple Choice
What is 50 C in Kelvin?
223 K
323 K
100 K
50 K
3
Multiple Choice
There are 65 liters of helium in a balloon at 35 K. If the temperature of the balloon is increased to 40 K, what will the new volume of the balloon be?
Which law would you use to solve this problem?
Charles' Law
Boyle's Law
4
Multiple Choice
Chlorine gas occupies a volume of 1.2 liters at 720 torr pressure. What volume will it occupy at 1 atm pressure?
Which law would you use to solve this problem?
Charles' Law
Boyle's Law
5
Multiple Choice
What is the formula for Lussac's Law?
T1P1=T2P2
T1V1/T2P2
P1/T1=P2/T2
T1/P1=T2/P2
6
Multiple Choice
At constant Volume,When Temperature increases then the Pressure must...
Increase
decrease
7
Multiple Choice
Pressure and temperature have what kind of relationship?
indirect
inverse
direct
synergistic
8
Multiple Choice
A gas is collected at 22.0˚C and 745.0 mm Hg. When the temperature is changed to 0˚C, what is the resulting pressure?
456 mmHg
521 mmHg
689 mmHg
725 mmHg
9
Multiple Choice
A container of gas is initially at 0.500 atm and 25˚C. What will the pressure be at 125˚C?
.419 atm
.668 atm
.765 atm
.974 atm
10
Multiple Choice
In ideal gases, volume and number of moles have which kind of relationship?
inverse
direct
indirect
obtuse
11
Multiple Choice
What units will be found in the variables of a Dalton's law problem?
kilopascals (kPa)
liters (L)
Kelvins (K)
moles (mol)
12
Multiple Choice
Dalton's law equation is used to find what?
the density of a pure gas
the total pressure of a pure gas
the total pressure of a gas mixture
the volume of a gas mixture
13
Multiple Choice
14
Multiple Choice
A tank of gas contains 50 kPa of helium, 50 kPa of argon, and 100 kPa of neon. What is the total pressure of the gas mixture?
100 kPa
50 kPa
200 L
200 kPa
15
Multiple Choice

left
right
they both have the same pressure
I don't know
Show answer
Auto Play
Slide 1 / 15
MULTIPLE CHOICE
Similar Resources on Wayground

12 questions
Simple Past Tense VS Present Perfect Tense
Presentation
•
10th Grade

10 questions
Making, accepting & declining invitations.
Presentation
•
10th Grade

11 questions
Cells - Prokaryotes vs. Eukaryotes
Presentation
•
10th Grade

10 questions
Molarity
Presentation
•
10th Grade

11 questions
SUMBER ENERGI
Presentation
•
10th Grade

11 questions
% PUREZA
Presentation
•
10th Grade

14 questions
Heating Curve Intro
Presentation
•
9th - 10th Grade

12 questions
Descriptive Text
Presentation
•
10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade