

Acid/Base and Reactions
Presentation
•
Chemistry, Science
•
8th - 11th Grade
•
Easy
Erin Hannan
Used 39+ times
FREE Resource
10 Slides • 8 Questions
1
Acid/Base and Reactions
Year 10 Chemistry
2
What we will learn about
definition of acids and bases
pH scale
indicators
neutralisation reactions
acid+ base
acid + carbonate
3
Definition of an acid
pH less than 7
turns blue litmus paper red
often sour
produces H+ ions in solution

4
Multiple Choice
Select the name of the following acid: HCl
Hydrochloric acid
Chlorous acid
Chloric acid
5
Definition of a base
pH greater than 7
turns red litmus paper blue
often bitter
produces OH+ ions in solution

6
pH scale
used to measure the acidity or basicity of a substance
takes the negative logarithmic value of H+ ions OR substances with more H+ ions will ne lower on the pH scale
Therefore, acids are low on the pH scale, and bases are high

7
Multiple Choice
Scale that measures how acidic or basic something is.
aH
bH
pH
Hp
8
Multiple Choice
Which pH is considered neutral?
1
3
7
14
9
Multiple Choice

The table shows the pH of several solutions. Which solution is the most acidic?
vinegar
milk
water
bleach
10
Multiple Choice
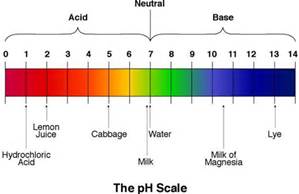
According to the pH range which substance is more acidic than lemon juice
Hydrochloric acid
cabbage
milk
11
pH indicators
we use pH indicators to test the pH of substances
their colour will change depending on the pH
gives a visual indicator, numerical pH values are given by technology
universal indicator is the most common

12
Acid + Base reactions

13
Acid + Base reactions
acid + base --> salt + water
neutralisation reaction
not always the case, but for now this is a good general equation
since you know the products, all you have to do is balance

14
Acid + carbonate reactions

15
Acid + carbonate reactions
acid + carbonate --> salt + water + carbon dioxide
again, a neutralisation
often used to see whether a substance is acidic
heating carbonates always decomposes the carbonate into its metal oxide + carbon dioxide
metal carbonate + heat --> metal oxide + CO2

16
Multiple Choice
Which salt will be produced if magnesium react with hydrochloric acid?
Magnesium oxide
Magnesium sulfate
Magnesium sulfide
Magnesium chloride
17
Multiple Choice
A neutralisation reaction is a reaction between
Acid and a Base
Acid and water
Base and water
None of the above
18
Multiple Choice
Complete the following reaction: Sulphuric acid + sodium carbonate -->
Carbon dioxide + water
calcium carbonate + water + carbon dioxide
Sodium sulphate + water + carbon dioxide
Sodium chloride + water + carbon dioxide
Acid/Base and Reactions
Year 10 Chemistry
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

14 questions
Ch. 5.1 - Models of the Atom
Presentation
•
9th - 11th Grade

12 questions
1-5: Solving Equations (Zero Product Property)
Presentation
•
8th - 10th Grade

14 questions
Trapezoids and Kites
Presentation
•
9th - 12th Grade

16 questions
Authors Purpose 8A (Mann)
Presentation
•
8th - 11th Grade

15 questions
AgApps Obj. 4.02 - Animal Biotechnology
Presentation
•
9th - 12th Grade

15 questions
LATIHAN TATABAHASA (KATA PENGUAT)
Presentation
•
9th - 12th Grade

13 questions
Mixture
Presentation
•
6th - 12th Grade

19 questions
Empirical and molecular formulas
Presentation
•
9th - 11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

19 questions
Review Set 3 - Moles & Chemical Formulas
Presentation
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Stoichiometry Practice
Quiz
•
10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Redox Reactions
Quiz
•
10th - 12th Grade

20 questions
pH and pOH Calculations
Quiz
•
10th - 11th Grade