

Chapter 3 and 4 Lecture
Presentation
•
Chemistry
•
University
•
Hard

Luis Bello
Used 2+ times
FREE Resource
45 Slides • 3 Questions
1
Chapter 3 and 4 Lecture
by Luis Bello
2
Chapter 3 Matter and Energy
Presenter Dr. Luis Bello

3
•3.3 Classifying Matter According to Its State: Solid, Liquid, and Gas
•3.4 Classifying Matter According to Its Composition: Elements, Compounds, and Mixtures
•3.5 Differences in Matter: Physical and Chemical Properties
•3.6 Changes in Matter: Physical and Chemical Changes
•3.7 Conservation of Mass: There Is No New Matter
•3.8 Energy
•3.9 Energy and Chemical and Physical Change
•3.1Temperature: Random Motion of Molecules and Atoms
•3.1Temperature Changes: Heat Capacity
•3.1Energy and Heat Capacity Calculations
4

5

6

7

8

9

10

11

12

13

14

15

16

17

18

19

20

21

22

23

24
Heat Capacity Definition
Specific heat is defined by the amount of heat needed to raise the temperature of 1 gram of a substance 1 degree Celsius (°C).
25

26

27
Chapter 4. Atoms and Elements
Presenter: Dr. Luis Bello
Source: Boundless Chemistry. Provided by: Boundless Learning. Located at: https://www.boundless.com/chemistry/textbooks/boundless-chemistry-textbook/. License: CC BY-SA: Attribution-ShareAlike

28
Chapter 4. Atoms and Elements
4.2 Indivisible: The Atomic Theory
4.3 The Nuclear Atom
4.4 The Properties of Protons, Neutrons, and Electrons
4.5 Elements: Defined by Their Numbers of Protons
4.6 Looking for Patterns: The Periodic Law and the Periodic Table
4.7 Ions: Losing and Gaining Electrons
4.8 Isotopes: When the Number of Neutrons Varies
4.9 Atomic Mass: The Average Mass of an Element’s Atoms
29
Early Ideas about Atoms
30
Multiple Choice
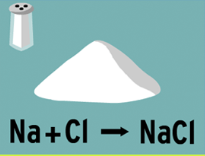
When sodium and chlorine combine to form sodium chloride, sodium chloride is the:
Originator
Reactant
Product
Produce
31
32

33

34

35

36

Periodic Table – Royal Society of Chemistry
You can open this webpage in a new tab.
37

38

39
Isotopes
40

41

42
Calculating Average Atomic Mass
43
The average atomic mass of an element is the sum of the masses of its isotopes, each multiplied by its natural abundance (the decimal associated with the percent of atoms of that element that are of a given isotope).
Average atomic mass = f1M1 + f2M2 + … + fnMn where f is the fraction representing the natural abundance of the isotope and M is the mass number (weight) of the isotope.
44

45

46
Multiple Choice
Do the following atoms belong to the same element? Explain Aluminum-27 An atom with 14 protons and 13 neutrons
Yes because they have the same number of protons
Yes because they have the same mass
No because they have different number of protons
No because they have different number of neutrons
47
Multiple Choice

How did Rutherford discover the proton?
Cathode Ray Tube
Gold Foil Experiment
Planetary Model
Plum Pudding Model
48

Chapter 3 and 4 Lecture
by Luis Bello
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

44 questions
7.05 & 7.06
Presentation
•
12th Grade

44 questions
PAST SIMPLE AND PAST PROGRESSIVE
Presentation
•
University

43 questions
Sepsis/Shock/MODS/AKI
Presentation
•
University

46 questions
Musical Instruments
Presentation
•
University

44 questions
FINTECH
Presentation
•
University

42 questions
Prefixes / sufixes
Presentation
•
University

41 questions
Application of Colligative Properties
Presentation
•
12th Grade

43 questions
ตัวดำเนินการบูลีน แก้ไขจากคุณ Narin Sintharom
Presentation
•
KG
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

40 questions
8th Grade Math Review
Quiz
•
8th Grade - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

10 questions
Would you rather...
Quiz
•
KG - University

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

14 questions
Superhero
Quiz
•
1st Grade - University