

FUNDAMENTAL EQUILIBRIUM CONCEPTS
Presentation
•
Chemistry
•
University
•
Medium

Luis Bello
Used 4+ times
FREE Resource
49 Slides • 12 Questions
1

2
Previous class
Chapter Outline
12.1 Chemical Reaction Rates
12.2 Factors Affecting Reaction Rates
12.3 Rate Laws
12.4 Integrated Rate Laws
12.5 Collision Theory
12.6 Reaction Mechanisms
12.7 Catalysis
3
Multiple Choice
An elementary reaction has only one activation-energy peak and one activated complex.
True
False
4
Multiple Choice

What is the molecularity of the elementary step shown here?
unimolecular
bimolecular
termolecular
5
Multiple Choice
A series of elementary steps that lead to an overall reaction is called a(n) ....
reaction mechanism
reaction combination
combustion reaction
exothermic reaction
6
Multiple Choice

How many elementary steps are in the reaction mechanism shown here?
1
2
3
7
Multiple Choice
What is collision theory?
Molecules must collide in the correct orientation with enough energy to bond.
Molecules need enough energy to collide and react.
Atoms constantly collide and react.
The minimum energy needed for atoms to react
8
Multiple Choice
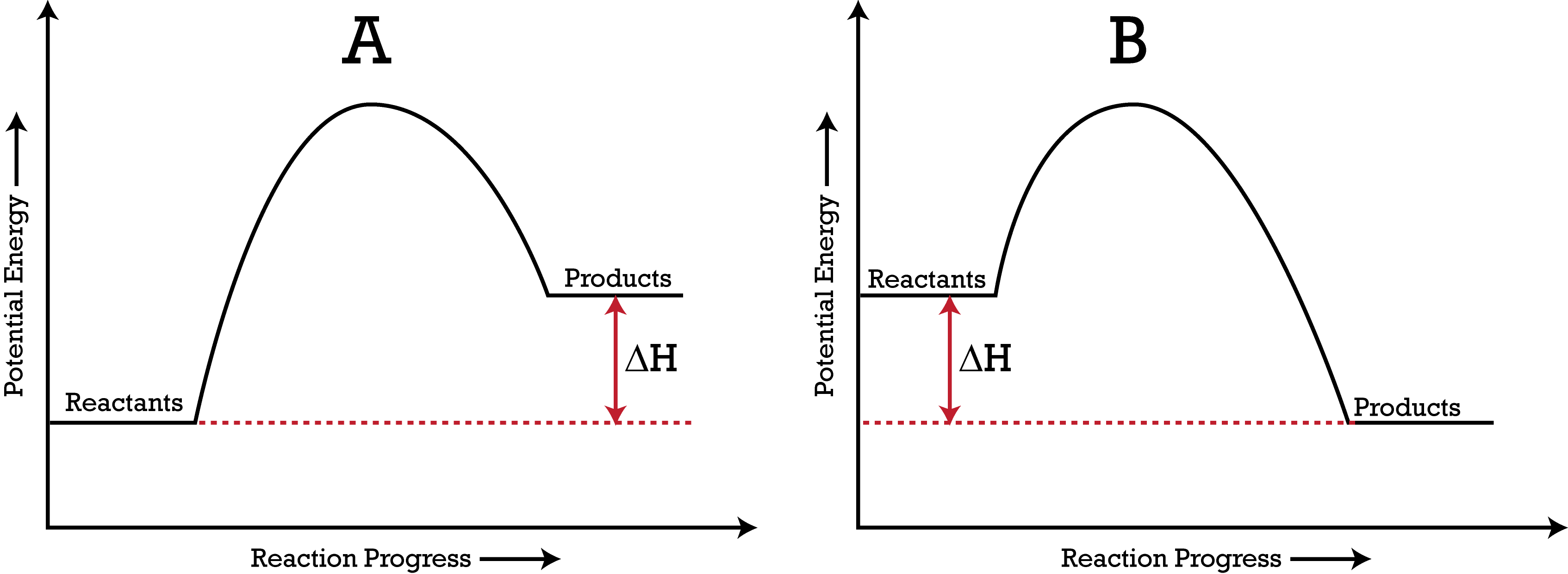
Which PE Diagram represents an endothermic reaction?
A
B
9
Multiple Choice
What factors effect rate of reaction?
Temperature, Concentration, Pressure and Energy
Surface Area, Concentration, Energy and Pressure
Temperature, Pressure, Concentration and Surface area
Pressure, Surface area, Density and Energy
10
Multiple Choice
X → products
Pure substance X decomposes according to the equation above. Which of the following graphs indicates that the rate of decomposition is second order in X ?
11
Multiple Choice
The following integrated rate law could be used to find the the concentration of a ________________ order reactant at a certain time.
[A] = - kt + [A]0
zero
first
second
12
Multiple Choice

The graphs shown in the diagram indicate that reactant A is ________________ order.
zero
first
second
13
Multiple Choice
How does a catalyst work in speeding up a reaction?
by lowering the activation energy or reaction
by giving them more energy
by making them more available
by increasing the activation energy or reaction
14
Multiple Choice
Catalysts may be in the state of ___________.
Solid and liquid only
Liquid and gases only
Solid and gases only
Solid, liquid or gases
15
16

17

18

19

20

21

22

23

24

25

26

27

28

29

30

31

32

33

34

35

36

37

38

39

40

41

42

43

44

45

46

47

48

49

50

51

52

53

54

55

56

57

58

59

60

61


Show answer
Auto Play
Slide 1 / 61
SLIDE
Similar Resources on Wayground

54 questions
GENERAL CHEMISTRY 2
Presentation
•
12th Grade

56 questions
Life 5 Unit 1 Lesson A
Presentation
•
University

56 questions
Neolithic Era
Presentation
•
KG - University

56 questions
Present Perfect and Present Perfect Continuous
Presentation
•
University

55 questions
Special Senses
Presentation
•
University

56 questions
Unit 1 Day 5 DHO 1:1 History of Healthcare slides
Presentation
•
KG - University

55 questions
Forms of Business Ownership
Presentation
•
University

52 questions
Moles and Stoichiometry-gkh
Presentation
•
University
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Chemistry

15 questions
LGBTQ Trivia
Quiz
•
University

36 questions
8th Grade US History STAAR Review
Quiz
•
KG - University

25 questions
5th Grade Science STAAR Review
Quiz
•
KG - University

16 questions
Parallel, Perpendicular, and Intersecting Lines
Quiz
•
KG - Professional Dev...

20 questions
5_Review_TEACHER
Quiz
•
University

10 questions
Applications of Quadratic Functions
Quiz
•
10th Grade - University

10 questions
Add & Subtract Mixed Numbers with Like Denominators
Quiz
•
KG - University

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...