

8th Grade Chemistry Pre-Test
Presentation
•
Chemistry
•
8th Grade
•
Medium
Jessica Harris
Used 4+ times
FREE Resource
1 Slide • 45 Questions
1

8th Grade Atomic Structure & Periodic Table Pre-Test
Mrs. Harris
2
Multiple Choice
3
Multiple Choice
4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
Most of the mass of an atom is found in the ______.
nucleus
proton
electron cloud
isotope
7
Multiple Choice
The smallest particle with characteristics of an element is known as the
Periodic Table
Atom
Proton
Electron
8
Multiple Choice
9
Multiple Choice

10
Multiple Choice
These are the SMALLEST subatomic particles
Proton
Neutron
Electron
11
Multiple Choice

12
Multiple Choice

13
Multiple Choice

Why is the overall charge of an atom neutral (0)?
They have the same number of protons and neutrons.
They have the same number of protons and electrons.
They have the same number of electrons and neutrons.
14
Multiple Choice
An atom with atomic number 6 would have how many protons
6
12
3
cannot be determined
15
Multiple Choice
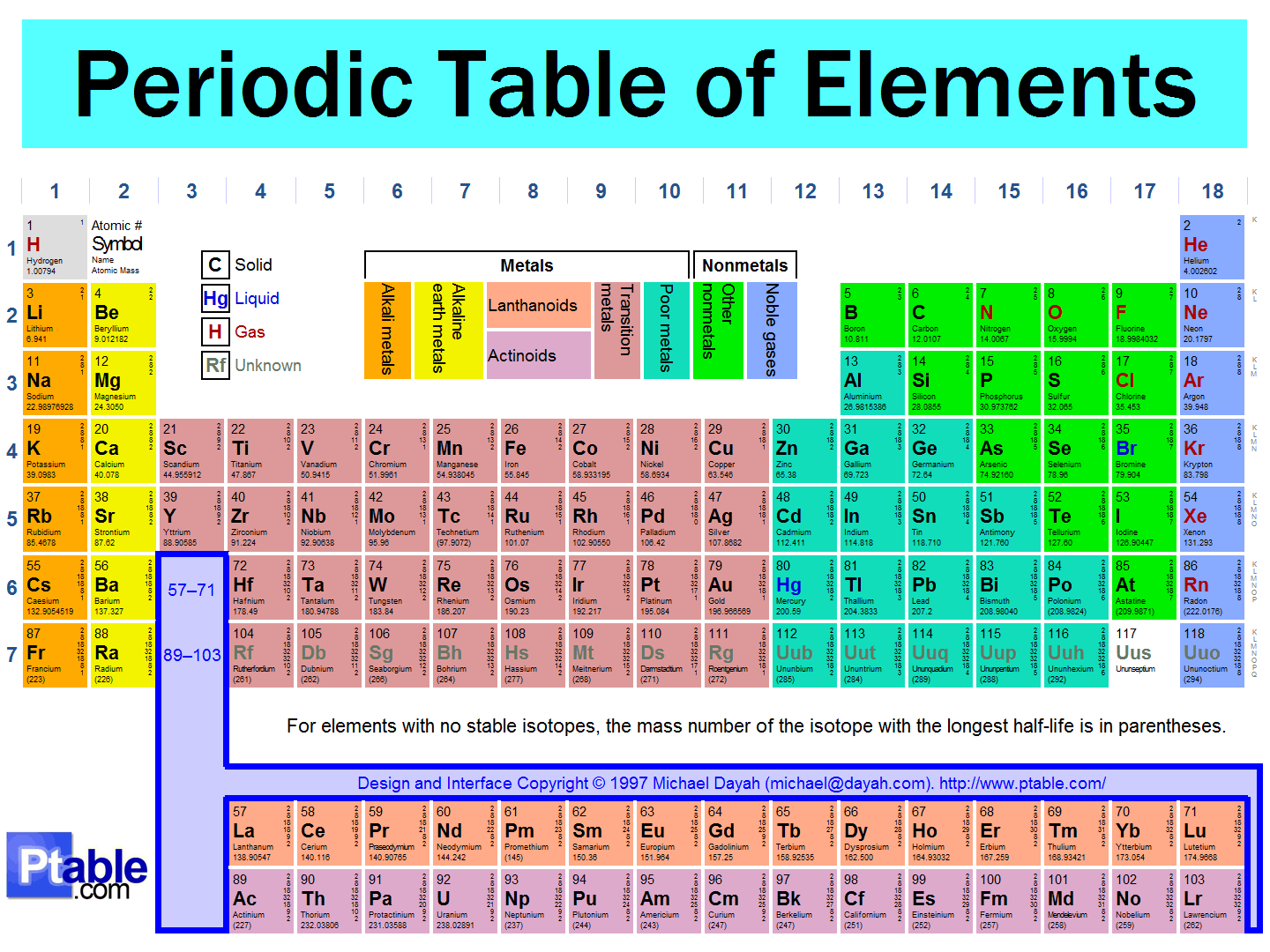
In the modern periodic table, elements are arranged by:
atomic mass
atomic number
valence electrons
number of isotopes
16
Multiple Choice

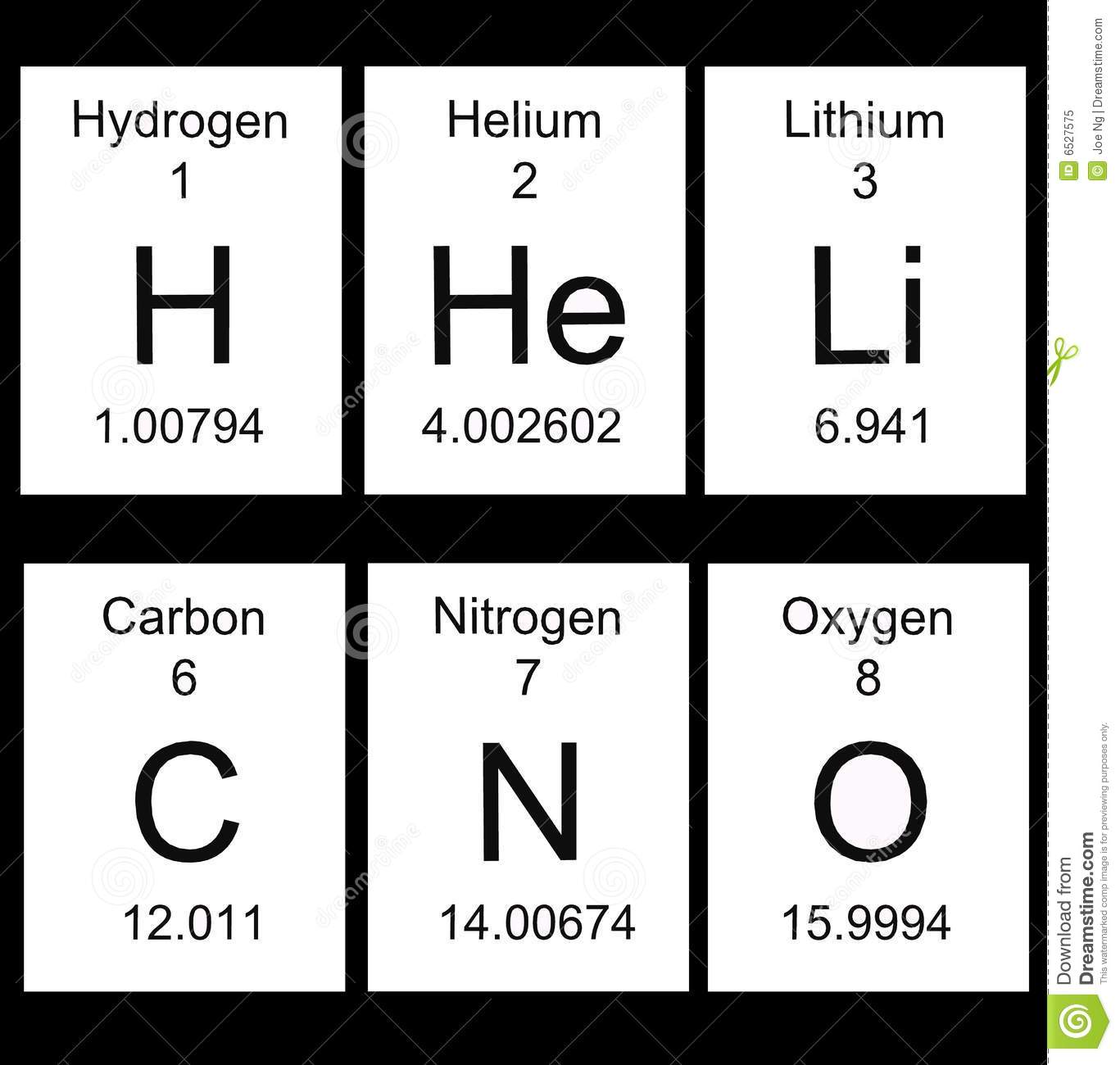
What does the 1.00794 stand for?
Hydrogen
atomic number
atomic mass
atomic explosion
17
Multiple Choice
If an atom has 10 protons, 10 neutrons, and 10 electrons, what is the mass of the atom?
10
20
30
18
Multiple Choice
True/False: Compounds are made of two or more different elements.
True
False
19
Multiple Choice
Most of the mass in an atom is concentrated in the...
electrons
nucleus
protons
empty space
20
Multiple Choice
An arrangement of elements in order of their atomic numbers such that elements with similar properties fall in the same column or group.
Atomic Number
Electron Cloud
Periodic Table
Nucleus
21
Multiple Choice
22
Multiple Choice
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice

27
Multiple Choice

28
Multiple Choice

29
Multiple Choice

30
Multiple Choice
MgCl2 --> Mg4+ Cl2
31
Multiple Choice
32
Multiple Choice
33
Multiple Choice
H2 + O2 -> H2O
34
Multiple Choice
H2 + O2 -> H2O
35
Multiple Choice

36
Multiple Choice
37
Multiple Choice

38
Multiple Choice
39
Multiple Choice
40
Multiple Choice
41
Multiple Choice
42
Multiple Choice
Covalent Bonds ______________ valence electrons.
give
take
keep
share
43
Multiple Choice
44
Multiple Choice
45
Multiple Choice
46
Multiple Choice

8th Grade Atomic Structure & Periodic Table Pre-Test
Mrs. Harris
Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

44 questions
Ecological Relationship
Presentation
•
8th Grade

40 questions
Area and Circumference of Circles
Presentation
•
7th - 8th Grade

39 questions
Intro to Slope
Presentation
•
8th Grade

39 questions
Simple Present Tense (8th Grade)
Presentation
•
8th Grade

38 questions
Structure and Properties of Matter
Presentation
•
9th Grade

39 questions
Holy Roman Empire and Feudalism
Presentation
•
8th Grade

38 questions
Tax Acts - Causes of the American Revolution
Presentation
•
8th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Chemistry

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade

20 questions
States of Matter
Quiz
•
8th Grade

41 questions
Atomic Structure and Periodic Table Unit Review
Quiz
•
8th Grade

20 questions
Chemical Reactions
Quiz
•
8th Grade

10 questions
Exploring Stoichiometry Concepts
Interactive video
•
6th - 10th Grade

10 questions
Exploring Solutions and Their Properties
Interactive video
•
6th - 10th Grade