

6.1 a/c/f: Intro to acid and base & Ka and Kb
Presentation
•
Science, Chemistry
•
10th Grade - University
•
Easy
Erin Hannan
Used 14+ times
FREE Resource
16 Slides • 11 Questions
1
Intro to acids/bases & Ka and Kb
Y12 Chem
2
Syllabus dot points
6.1 a investigate the correct IUPAC nomenclature and properties of common inorganic acids and bases
c predict the products of acid reactions and write balanced equations to represent:
– acids and bases
– acids and carbonates
– acids and metals
f explore the changes in definitions and models of an acid and a base over time to explain the limitations of each model, including but not limited to:
– Arrhenius’ theory
– Brønsted–Lowry theory
3
What we will cover today...
what is an acid and what is a base?
how to name acids (and bases)
conjugate acid/base pairs
Ka and Kb defintions
4
What is an acid and a base?
several definitions
developed over time - used to be based on simpler observations (litmus tests, reactions)
there are several theories/definitions:
Arrhenius
Bronsted-Lowry
Lewis

5
Open Ended
Do you remember any of these definitions of an acid or a base?
6
Arrhenius
Acids = any substance that dissociates (breaks apart) and produces H+ ions
Bases = dissociate in water to produce hydroxide ions

7
Bronsted-Lowry
Acids = proton donors
Bases = proton acceptors
an acid base reaction will involve an exchange of protons from an acid to a base
usually in the form of an H+ ion
this expands acid/base reactions out a solution

8
Lewis
Acid = substance that accepts a pair of electrons
Base = substance that donates a pair of electrons


9
Multiple Choice
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice

13
Properties of acids and bases


14
Naming Acids

15
Reactions of acids
acid + base -> salt + water
acid + carbonate -> salt + carbon dioxide + water
acid + metal -> salt + hydrogen gas



16
Multiple Choice
Which of the following chemical reactions represents an acid-base reaction?
HBr + KOH → KBr + H2O
ZnCl2 + MgSO4 → ZnSO4 + MgCl2
H2SO4 + CaCl2 → CaSO4 + HCl
NH4OH + KCl → KOH + NH4Cl
17
Multiple Choice
Identify the salt in the following equation:
HNO3 + Zn(OH)2 → Zn(NO3)2 + H2O
HNO3
Zn(OH)2
Zn(NO3)2
H2O
18
Multiple Choice
19
Multiple Choice
Hydrochloric acid + magnesium hydroxide -->
20
Conjugate acid-base pair
two molecules or ions that differ by one proton (H+ ion)
we consider this relationship in terms of the reactions that occur
e.g. HCl and Cl- are a conjugate acid-base pair. Cl- is formed from HCl by the loss of a single proton, and is the conjugate base of HCl.

21
Multiple Choice
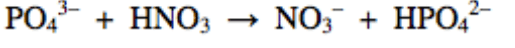
22
Multiple Choice
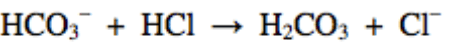
Cl-
23
Strong vs Weak Bases
strong ionises completely in a solution
weak only partially ionises
therefore, the ionisation of a weak acid or bases is a reversible reaction - as it never fully goes to completion
weak acids = HA
conjugate base = A-
weak base = B
conjugate acid = BH+
24
Ka and Kb
acid dissociation constant
base dissociation constant
describes the degree of ionisation of an acid or a base - and therefore indicate the strength of the acid or base
25
Ka
general equation for the ionisation of a weak acid in water
HA is the acid (donates a proton)
A- is the conjugate base
H3O could be written as H+, however, free H+ doesn't exist in solution
the larger the Ka, the stronger the acid and the higher the H+ concentration at equilibrium


26
Kb
weak bases react with water and produce hydroxide ions
In general reaction:
B = the parent base
BH = conjugate acid
the larger the Kb, the stronger the base and the higher the OH- concentration at equilibrium


27
Next Few Lessons
Acid/base general reactions
Indicators
Enthalpy of Neutralisation
Intro to acids/bases & Ka and Kb
Y12 Chem
Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

22 questions
Molarity Calculations
Presentation
•
10th - 12th Grade

25 questions
Physical and Chemical Changes Review
Presentation
•
8th Grade

20 questions
Reading Strategies: Context Clues and LA Terms
Presentation
•
10th - 12th Grade

21 questions
Charles's Law and the Combined Gas Law
Presentation
•
11th - 12th Grade

20 questions
Exponent Rules
Presentation
•
9th - 12th Grade

22 questions
Similar Figures, find scale factor and solve for missing sides
Presentation
•
KG

21 questions
Cover Letters and Resumes
Presentation
•
9th - 12th Grade

22 questions
Chp. 1 Video 1
Presentation
•
11th Grade - University
Popular Resources on Wayground

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

36 questions
6th Grade Math STAAR Review
Quiz
•
6th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

15 questions
Chemical Reactions (Types of Chemical Reactions)
Interactive video
•
11th Grade

10 questions
Exploring the Layers of the Atmosphere
Interactive video
•
6th - 10th Grade

10 questions
Exploring Ecological Levels: Organism to Ecosystem
Interactive video
•
6th - 10th Grade

10 questions
Exploring Chemical and Physical Changes
Interactive video
•
6th - 10th Grade

20 questions
Cladograms and Phylogenetic Trees
Quiz
•
9th - 12th Grade

9 questions
Phylogeny
Quiz
•
10th Grade

12 questions
Cellular Respiration
Interactive video
•
11th Grade