

Grade 6 Term 2 Revision 21-22
Presentation
•
Science
•
6th Grade
•
Easy
Tanzeel Haq
Used 12+ times
FREE Resource
32 Slides • 52 Questions
1
Grade 6 Term 2 Revision
By Tanzeel Haq
2

3

4
Multiple Choice
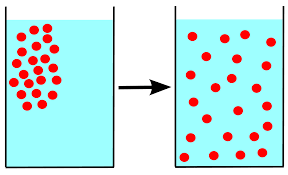
The picture below is an example of _________________.
osmosis
isotonic
diffusion
active transport
5
Multiple Choice
How do the particles move in diffusion?
From a region of low concentration to a region of high concentration
From a region of high concentration to a region of low concentration
From a region of low concentration to a region of low concentration
From a region of high concentration to a region of high concentration
6
Multiple Choice
How long does diffusion last?
Until the concentration of particles in the system is equal
(Select this answer and I swear i'll make it harder for you in the assessment task)
Until a region in the system has a greater concentration of particles than the other
Until the particles disappear from the system
7

8
Multiple Choice

Thermal expansion occurs when particles of matter move farther apart as temperature increase.
true
false
9
Multiple Choice
10

11
Draw
Draw a particle diagram to show WHat happens when a gas expnands.
12

13
Open Ended
WHat Happens to the solid metal when the Temperatre is increased?
14

15
Multiple Choice
Which statement best describes the importance of joints in the roadways of bridges?
Joints strengthen the roadway to support cars and trucks
Joints allow the roadway to expand and contract with changing outside temperatures
Joints allow the roadway to expand and contract as cars put force on the bridge
Joints allow the bridge to be built in smaller, easier to assemble pieces.
16
Multiple Choice
Choose the picture depicting the solid at the highest temperature
17
Multiple Choice
Do you think thermal expansion affects all states of matter or just one?
All States of Matter
Just Solids
Just Liquids
Just Gases
18

19
Multiple Choice
20

21
Multiple Choice
A camera which produces images with different colors based on the heat of an object is
Digital camera
Thermogram
Thermometer
Thermos camera
22
Multiple Choice
The object that provides energy for the energy transfer is called __________.
source object
receiver object
empty object
energy object
23
Multiple Choice
The object that gains energy from the energy transfer is called _________.
source object
receiver object
empty object
energy object
24
Multiple Choice
When you are heating food in a microwave oven, what object is the food?
source object
receiver object
25

26
Multiple Choice

Conduction
Convection
Radiation
27
Multiple Choice
How does heat energy reach the Earth from the Sun
Convection
Radiation
Conduction
Insulation
28
Multiple Choice

Conduction
Convection
Radiation
29
Multiple Choice

Conduction
Convection
Radiation
30
Multiple Choice

31

32
Multiple Choice
33
Multiple Choice
34

35
Open Ended
Calculate the Change in Temperature
Initial temp = 40 C
Final temp = 65 C
36

37
Draw
Draw how particles move in Solid liquid and gas
38

39
Multiple Choice
H2SO4
40
Multiple Choice
41
Multiple Choice
Carbon is considered an element, while carbon dioxide is considered a compound. This is because carbon dioxide is ---
a gas at room temperature.
made of two different elements.
given off by green plants.
useful to human beings
42

43
Multiple Choice
44
Multiple Choice
45

46

47
Multiple Choice
In CHARLES'S LAW, volume and temperature have a ______ proportionality.
direct
inverse
linear
exponential
48
Multiple Choice

In CHARLES' LAW, what happens to temperature of a gas when the volume doubles?
The temperature also doubles.
The temperature decreases to half of its original volume.
The temperature doesn't change.
The temperature quadruples.
49

50

51

52
Multiple Choice
53
Multiple Choice

54

55
Multiple Choice
What causes air pressure?
force of collisions from air particles
air particles increasing in size
increasing the mass of each air particle
air particles changing state
56
Multiple Choice

What type of relationship is there between pressure and temperature?
inversely proportional
proportional
no relationship
57
Multiple Choice

What concept on pressure does the image show?
adding particles increases pressure
more mass increases pressure
larger particles increases pressure
more space between particles increases pressure
58
Multiple Choice
Which cooking device will allow you to heat up water hotter than it's boiling point?
skillet
pressure cooker
crock pot
pasta pan
59

60
Multiple Choice
Non-metals are insulators, this means electricity:
Can flow through them
Cannot flow through them
61
Multiple Choice
Hydrogen, Helium, and Fluorine are all listed as -
semi-metals
non-metals
metalloids
metals
62

63
Multiple Choice

The following properties are all characteristics of ionic compounds EXCEPT
high melting and boiling points
soft
crystal lattice structure
conduct electricity when dissolved in water
64
Multiple Choice

Covalent compound are _________ conductors of electricity and thermal energy.
good
poor
neither
65

66

67
Multiple Choice
68
Multiple Choice
69
Multiple Choice

70
Multiple Choice
71

72

73

74
Draw
Mark the place in the graph where the Kinetic energy will be highest
75
Draw
Mark the place in the graph where the Potential energy will be highest
76

77

78

79

80
Multiple Choice
No bubbles are formed.
boiling
evaporation
81
Multiple Choice
Can happen at any temperature
boiling
evaporation
82
Multiple Choice
Bubbles form
boiling
evaporation
83
Multiple Choice
Happens only at the surface of the liquid
boiling
evaporation
84
Multiple Choice
Evaporation can happen even when a liquid is not boiling.
true
false
Grade 6 Term 2 Revision
By Tanzeel Haq
Show answer
Auto Play
Slide 1 / 84
SLIDE
Similar Resources on Wayground

78 questions
UK, Germany, and Russia Impact of Location, Climate, and Resourc
Presentation
•
6th Grade

75 questions
Nature and Changing Ecosystems
Presentation
•
6th Grade

80 questions
Ecosystems - Food Chains/Webs
Presentation
•
6th - 8th Grade

78 questions
EC A2 8.3
Presentation
•
6th Grade

81 questions
Theory of Colour
Presentation
•
6th Grade

76 questions
Science Flashcards
Presentation
•
5th Grade

76 questions
Iceland Adventure: Land of Fire and Ice
Presentation
•
6th Grade

74 questions
Identifying DNA as the genetic material
Presentation
•
KG
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

20 questions
Rocks and The Rock Cycle
Quiz
•
6th Grade

20 questions
Moon Phases and Eclipses
Quiz
•
6th - 8th Grade

20 questions
Human Body Systems
Quiz
•
6th Grade

22 questions
6th & 8th Grade Science Material SOL Review
Quiz
•
6th - 8th Grade

15 questions
Punnett Squares
Quiz
•
6th Grade

20 questions
Cells! Cell Theory and Characteristics of Eukaryotes/Prokaryotes
Quiz
•
6th Grade

35 questions
Amplify Weather Patterns Unit Review
Quiz
•
6th Grade