

Chem. Rev. Chapter 5 Chemical reaction
Presentation
•
Chemistry
•
9th - 10th Grade
•
Hard
Michelle Zou
FREE Resource
17 Slides • 31 Questions
1
Chem. Rev. Chapter 5 Chemical reaction
By Michelle Zou
2

3
Multiple Choice
What is the left part of a chemical equation called?
2H2 + O2 ---> 2H2O
Reactants
Yields
Products
Chemical equation
4
Multiple Choice
What is the right part of a chemical equation called?
2H2 + O2 ---> 2H2O
Reactants
Yields
Products
Chemical Equation
5

6
Multiple Choice

The Law of Conservation of Mass states Matter is NEITHER created nor destroyed.
True
False
7
TRUE!
The Law states matter is neither created nor destroyed as seen in this image. We started with 100g and when we broke apart the elements they STILL equal 100g of mass.

8
Multiple Choice
9
Multiple Choice
10

11

12
Multiple Choice

13

14
What is a molecule?

15

16

17

18

19
Multiple Choice
20

21

22
LET'S TAKE IT STEP BY STEP AND PRACTICE
23
Multiple Choice
24
Multiple Choice

How many oxygen atoms are in this chemical formula?
6 oxygen atoms
2 oxygen atoms
3 oxygen atoms
4 oxygen atoms
25
Multiple Choice
26
Multiple Choice
Which of the following has the most molecules of O?
2Mg3(PO4)2
4AlPO4
3Al(NO3)3
15KOH
27
Multiple Choice
Which of the following equations are correctly balanced?
12CO2 + H2O ---> C6H12O6 + O2
CO2 + H2O ---> 3C6H12O6 + O2
CO2 + 9H2O ---> C6H12O6 + O2
6CO2 + 6H2O ---> C6H12O6 + 6O2
28
Multiple Choice
Balance this equation:
HgO ---> 2Hg + O2
It is balanced.
2HgO ---> 2Hg + O2
2HgO ---> Hg + O2
HgO ---> Hg + 2O2
29
Multiple Choice
Mg + __HCl ---> MgCl2 + H2
30
Multiple Choice
Balance the following equation:
___ H2+ ___ O2→___ H2O
1, 1, 3
2, 2, 2
1, 2, 1
2,1, 2
31
Multiple Choice

Balance the following equation:
__CO + __Fe2O3--> __Fe + __CO2
3,1 --> 2,3
3,2 --> 1,1
3,2 --> 2,4
3,2 --> 2,1
32
Multiple Choice

Balance the following equation:
__Al + __O2--> __Al2O3
4,1 --> 2
4,3 --> 4
3,4 --> 1
4,3 --> 2
33
Multiple Choice
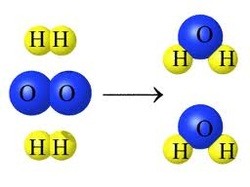
Which equation supports this model?
2H2 + O2 → 2H2O
H2 + O2 → 2H2O
2H2O2 → 2H2O
2H2 + O2 → H2O
34
Relative Formula Mass
A measure (still not the exact mass) of a molecule, using the relative atomic mass on the Periodic Table.

Sometimes called relative molecular mass, molar mass etc. All using the same procedure.
35
Multiple Choice
Name this compound:
CO
Carbon Oxide
Carbon Oxygen
Dicarbon dioxide
Carbon Monoxide
36
Multiple Choice
Calculate the relative formula mass for Al(OH)3
78.0
72
28
25
37
Multiple Choice
What is the relative formula mass of CO2?
38
Multiple Choice
39
Multiple Choice
What is the unit of relative formula mass?
40
Multiple Choice
41
Multiple Choice
What is the general formula for the computation of percentage composition of the given element?
% mass of element = mass of element in the compound divided by mass of the compound times 100
% mass of element = mass of element in the compound divided by mass of the compound
% mass of element = mass of element in the compound divided by 100
% mass of element = mass of element in the compound
42
Multiple Choice
What is the percent by mass of magnesium in MgO?
20%
40%
50%
60%
43
Multiple Choice
What is the percent by mass of chlorine in NaCl?
39%
61%
35%
35%
44
Fill in the Blank
45
Multiple Choice
In a chemical reaction, 4 grams of sodium must combine with how many grams of chlorine to produce 10 grams of table salt?
4 grams
6 grams
8 grams
10 grams
46
Multiple Choice
2Na + 2H2O → 2NaOH+ H2
How many grams of hydrogen are produced if 120 g of Na are available?
5.27 g
2.6 g
690 g
45 g
47
Multiple Choice
2H2 + O2 → 2H2O How many grams of water can be produced if 8 gram of O2 are used?
4.5 g
9 g
18 g
8 g
48
Well done MBSer!
Chem. Rev. Chapter 5 Chemical reaction
By Michelle Zou
Show answer
Auto Play
Slide 1 / 48
SLIDE
Similar Resources on Wayground

45 questions
Lab Safety
Presentation
•
10th Grade

38 questions
Structure and Properties of Matter
Presentation
•
9th Grade

45 questions
Physical Characteristics of the Middle East
Presentation
•
9th Grade

43 questions
Sulphur - full topic (review + 16/11/2020 lesson)
Presentation
•
10th Grade

39 questions
Genetics terms part 1
Presentation
•
9th - 10th Grade

44 questions
Periodic Trends
Presentation
•
10th Grade

40 questions
Cell terms part 1
Presentation
•
9th - 10th Grade

42 questions
All About Venn Diagrams
Presentation
•
10th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

15 questions
Hargrett House Quiz: Community & Service
Quiz
•
5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

15 questions
Equivalent Fractions
Quiz
•
4th Grade
Discover more resources for Chemistry

20 questions
Acids and Bases
Quiz
•
10th Grade

30 questions
Element compound mixture review quiz
Quiz
•
10th - 12th Grade

10 questions
Determining Protons, Neutrons, and Electrons from the Periodic Table
Interactive video
•
6th - 10th Grade

10 questions
Identifying Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

20 questions
U4 Reaction Types
Quiz
•
10th - 12th Grade

10 questions
Exploring Types of Chemical Reactions
Interactive video
•
6th - 10th Grade

24 questions
Ecology Quiz Review
Quiz
•
9th Grade

20 questions
Elements, Compounds, and Mixtures
Quiz
•
10th Grade