

Matter Unit Powerpoint Review
Presentation
•
Science
•
6th - 8th Grade
•
Medium
+4
Standards-aligned
Erin Jameson
Used 31+ times
FREE Resource
28 Slides • 33 Questions
1
Matter Unit Powerpoint Review
By Erin Jameson
2

3

4

5

6
Multiple Choice
What is the smallest piece of matter that still has properties of that matter?
element
atom
proton
7
Multiple Choice
8
Multiple Choice
Negatively charged particle
proton
neutron
electron
9
Multiple Choice
10

11

12

13

14

15

16

17
Multiple Choice
A(n) ______________ is formed when two or more elements chemically combine.
Compound
Element
Mixture
18
Multiple Choice
A ___________ is a combination of many different elements not chemically combined.
Element
Compound
Mixture
19
Multiple Choice

20
Multiple Choice
Which of the following is classified as a pure substance?
Lemonade
Fruit punch
ketchup
Hydrogen
21
Multiple Choice
It is a combination of two or more substances in
which the composition is uniform throughout the
mixture.
Heterogeneous mixture
Homogeneous mixture
Solvation
Solution
22
Multiple Choice
Which of the following describes a substance that has been physically combined and is NOT uniform throughout (you see the DIFFERENT parts that make it up)?
Homogeneous Mixture
Compound
Heterogeneous mixture
element
23
Multiple Choice
This method separates substances due to differences in the sizes of their particles.
Filtration
Evaporation
Distillation
Chromatography
24

25
Multiple Choice

Element
Compound
Mixture
26
Multiple Choice

Element
Compound
Mixture
27
Multiple Choice

Element
Compound
Mixture
28

29
Multiple Choice
The number of electrons in an atom is equal to the...
Protons and Neutrons
Protons
Atomic Mass
Neutrons
30
Multiple Choice
The formula used to calculate the number of neutrons is:
Mass Number - Atomic Number
Protons + Electrons
Atomic Number - Mass Number
Protons x2
31
Multiple Choice
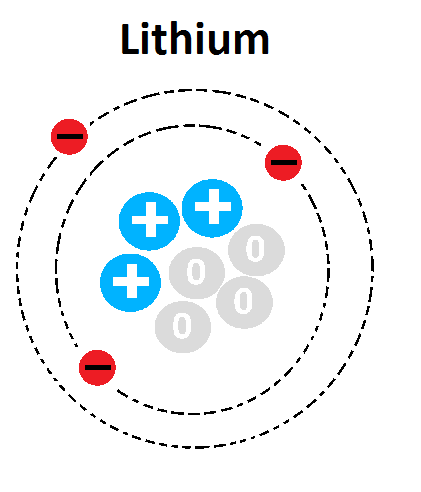
32

33

34

35

36

37

38

39
Multiple Choice

40
Multiple Choice
These two elements have similar chemical properties to Barium (Ba).
Ca and Ra, because they are in the same group.
Cs and La, because they are in the same period number.
Ca and Y, because they are 90 degree angle.
41
Multiple Choice
Which group will not combine with other elements because they have a full outer shell of electrons?
Alkaline Earth Metals
Alkali Metals
Noble Gases
Halogens
42
Multiple Choice
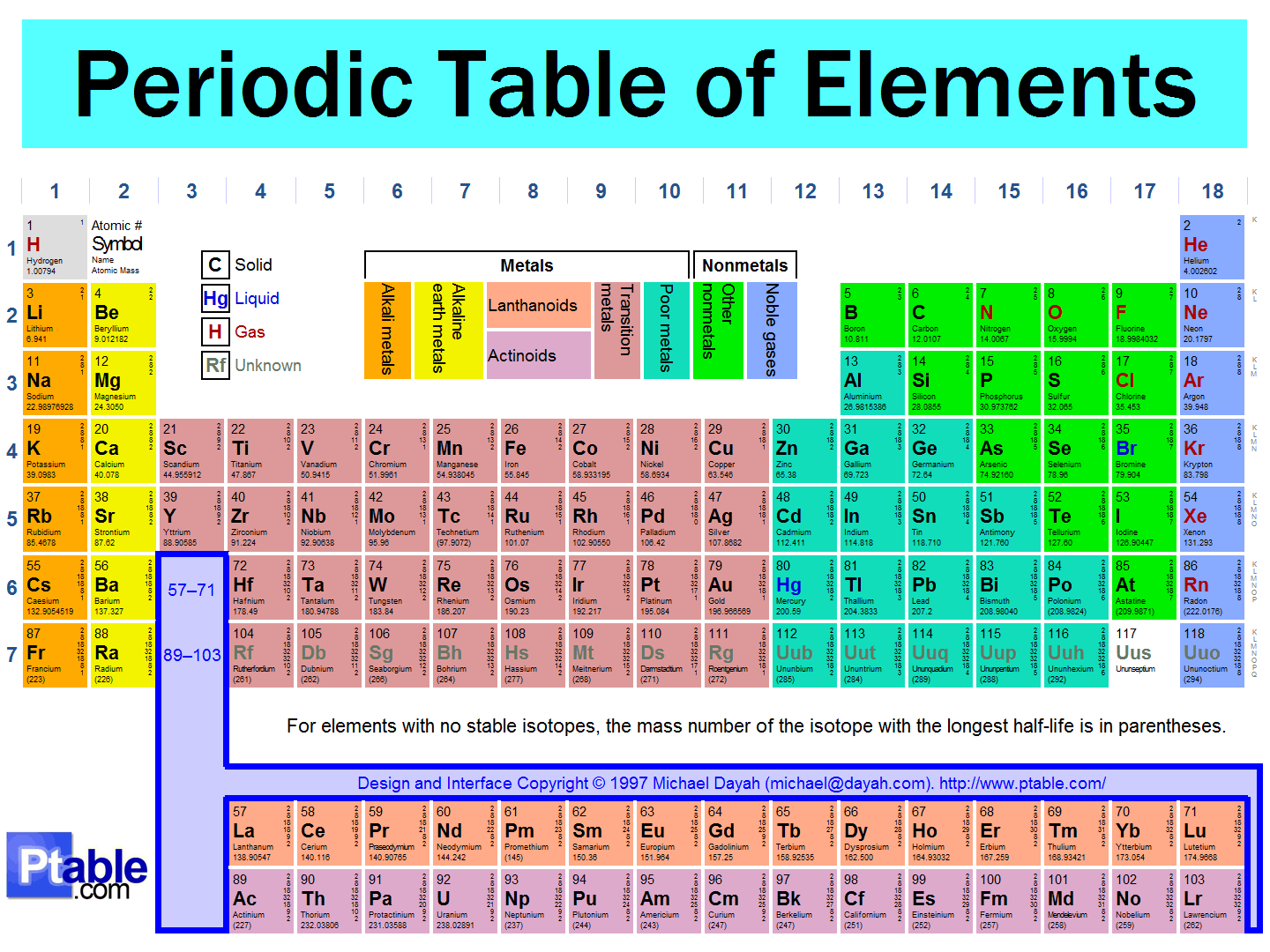
43
Multiple Choice
an arrangement of the elements according to their atomic numbers so that elements with similar properties are in the same column and properties repeat from row to row
Periodic Table of Elements
Atomic Theory
Isotopes
Subatomic Particles
44

45

46
Multiple Choice
47
Multiple Choice
Boiling point: Chemical or Physical Property
48

49

50

51
Multiple Choice
52
Multiple Choice

Which is the correct description of photosynthesis
Photosynthesis is a chemical reaction because the atoms of the molecules get rearranged to form new molecules
Photosynthesis is a chemical reaction because the molecules stay the same before and after the change
Photosynthesis is a physical change because nature makes it happen
Photosynthesis is a physical change because the atoms stay the same
53
Multiple Choice

Baking a cake is an example of a
Chemical change
Physical chage
54
Multiple Choice
55

56

57
Multiple Choice

The law of conservation of mass says substances can neither be _______ nor ___________.
Built; torn down
Blended together; separated
Created; destroyed
Condensed; extracted
58
Multiple Choice
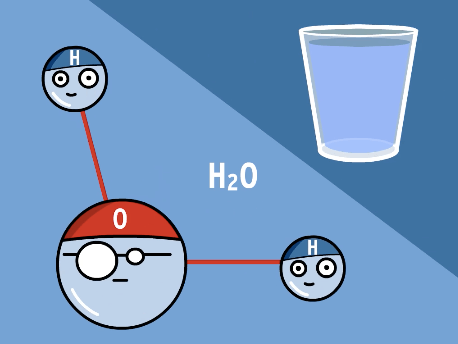
4 grams of hydrogen and 32 grams of oxygen will combine to form:
36 grams of water
28 grams of hydroxide
32 grams of oxygen
36 grams of deuterium
59
Multiple Choice
60
Multiple Choice
According to the Law of Conservation of Mass, what happens to the wood and oxygen when it is burned?
The wood disappears.
It stays the same chemically.
The atoms in the wood and oxygen rearrange and form smoke and ash.
Nothing.
61
Multiple Choice
Balance the following equation:
___ Mg+ ___ O2→___ MgO
1, 1, 3
2, 1, 2
1, 2, 1
2, 2, 2
Matter Unit Powerpoint Review
By Erin Jameson
Show answer
Auto Play
Slide 1 / 61
SLIDE
Similar Resources on Wayground

54 questions
Chapter 24: Stars, Galaxies, and the Universe
Presentation
•
6th - 8th Grade

56 questions
Properties of Minerals Guided Lesson
Presentation
•
6th - 8th Grade

56 questions
LEGO My Element!
Presentation
•
6th - 8th Grade

58 questions
Mitosis vs Meiosis
Presentation
•
7th Grade

52 questions
Forms of Energy
Presentation
•
6th - 7th Grade

55 questions
Mid-term Science Review
Presentation
•
6th - 8th Grade

57 questions
SCOP Mod 1 Lesson Edit for Share
Presentation
•
KG - 8th Grade

55 questions
7th- Nervous System 4.2 (The Brain)
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

16 questions
Photosynthesis and Cellular Respiration Review
Quiz
•
7th Grade

20 questions
Abiotic & Biotic Factors
Quiz
•
7th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade