

Benchmark 4 review day 1 (Unit 10 thermo,K&E)
Presentation
•
Chemistry
•
10th Grade - University
•
Medium
+5
Standards-aligned
Katie Smit
Used 6+ times
FREE Resource
27 Slides • 30 Questions
1
Wednesday 5/11- Benchmark review- Unit 10
Announcements/Reminders
Agenda:
12 class DAYS until final!
Thurs 5/9- English EOC, 1st &2nd block
Mon 5/13-Benchmark #4
5/22- no school- Biology EOC
5/23- USH EOC
5/28- final exam (1st&2nd periods)
Unit 10 review
Bellwork:
2

3

4

5

6

7

8

9

10

11

12

13

14
Multiple Choice
Can energy be created?
Yes
No
15
Multiple Choice
Which of the following is not a form of energy?
Thermal (heat)
temperature
light
16
Multiple Choice

Heat flows from _______ to ______ objects
cooler to warmer
warmer to warmer
warmer to cooler
cooler to cooler
17
Multiple Choice

Phase changes: ice to water to steam
endothermic
exothermic
18
Multiple Choice

Phase changes: steam to water to ice
endothermic
exothermic
19
Multiple Select
Select all the processes that are exothermic. The system is underlined in each example.
Your hand gets cold when you touch ice.
The ice gets warmer when you touch it.
Water boils in a kettle being heated on a stove.
Water vapor condenses on a cold pipe.
Ice cream melts.
20
Multiple Select
Select all the processes that are endothermic. The system is underlined in each example.
Your hand gets cold when you touch ice.
The ice gets warmer when you touch it.
Water boils in a kettle being heated on a stove.
Water vapor condenses on a cold pipe.
Ice cream melts.
21
Multiple Choice
22
Multiple Choice
O2(g) + 2 H2 (g) → 2 H2O (g) + Energy
endothermic
exothermic
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice

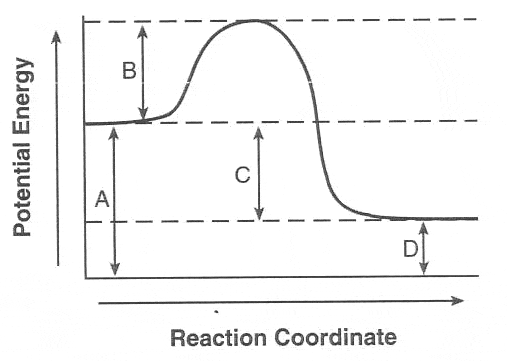
What is the Heat of Reaction ΔH for this reaction?
-200 KJ
200 KJ
400 KJ
-300 KJ
26
Multiple Choice

How much activation energy is needed for this reaction?
300 KJ
500 KJ
400 KJ
100 KJ
27
Multiple Choice

What type of reaction is this?
Exothermic
Endothermic
Energy Producing
No way to tell
28
Multiple Choice
What kind of reaction is this: 2KI + MgCl2 + 200KJ ----> 2KCl + MgI2
Endothermic
Exothermic
29
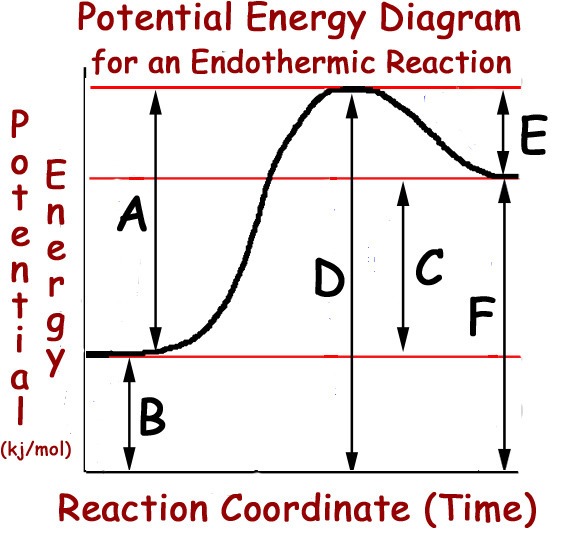
Multiple Choice
30
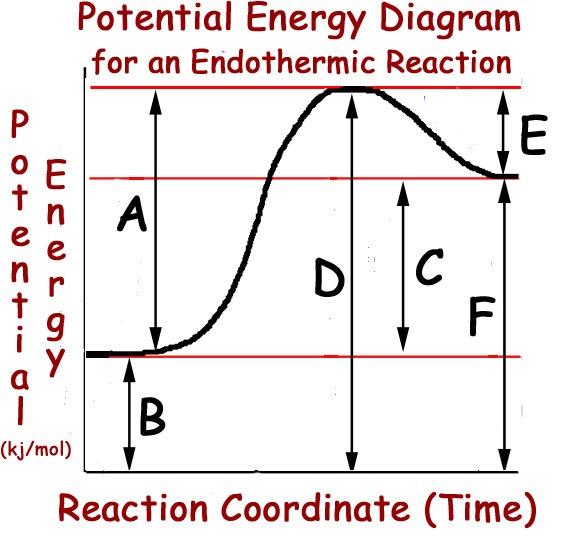
Multiple Choice
31
Multiple Choice
32
Multiple Choice

33
Multiple Choice

34

35

36
nIonic solutions have faster reactions than molecule compounds. (bonding)
néTemp. é Rate
né conc. érate
né surface area é rate
né Pressure é rate, ê P ê rate
nCatalysts speed up reactions.
Quick review- Factors affecting rate
37

38
Multiple Choice

This symbol indicates that a reaction is _____
reversible
irreversible
39

→
40
Multiple Choice
41
Multiple Choice
42

43

44

45

46

47
Concentration increase shift away from increase
Concentration decrease shift toward decrease
🡹 pressure shifts in direction of fewer gas molecules.
🡻 pressure shifts in direction of more gas molecules
🡹 temperature favors endothermic reaction
Shift away from heat
🡻 temperature favors exothermic reaction
Shift towards heat
Equilibrium shifts due to stresses:
48
Example- The Haber process (making ammonia NH3)
N2 (g) + 3 H2 (g) ⮀ 2 NH3 (g) + heat
🡹 [N2]
🡻 [H2]
🡹 [NH3]
🡻 [NH3]
🡹 pressure
🡻 pressure
🡹 temperature
🡻 temperature
49

50

51
Multiple Choice
52
Multiple Choice
(🡹 pressure shifts in direction of fewer gas molecules)
(🡻 pressure shifts in direction of more gas molecules )
N2(g) + 3H2(g) ↔ 2 NH3(g) + heat
Increasing the pressure will cause the equilibrium to:
Shift right
Shift left
Have no change
Speed up
53
Multiple Choice
🡹 temperature favors endothermic reaction Shift away from heat 🡻 temperature favors exothermic reaction Shift towards heat
N2(g) + 3H2(g) ↔ 2 NH3(g) + heat
Increasing the temperature will cause the equilibrium to:
Shift right
Shift left
Have no change
Speed up
54
Multiple Choice
(Concentration increase shift away from increase)
(Concentration decrease shift toward decrease)
N2(g) + 3H2(g) ↔ 2 NH3(g) + heat
Decreasing H2 will cause the equilibrium to:
Shift right
Shift left
Have no change
Speed up
55
Multiple Choice
(Concentration increase shift away from increase)
(Concentration decrease shift toward decrease)
N2(g) + 3H2(g) ↔ 2 NH3(g) + heat
Removing N2 will cause the equilibrium to:
Shift right
Shift left
Have no change
Speed up
56
Multiple Choice
(Concentration increase shift away from increase)
(Concentration decrease shift toward decrease)
N2(g) + 3H2(g) ↔ 2 NH3(g) + heat
Increasing H2 will cause the equilibrium to:
Shift right
Shift left
Have no change
Speed up
57
Multiple Choice
SO2(g) + O2(g) <−> SO3(g)
If the concentration of SO2(g) is increased, the equilibrium of the reaction will ___________.
Wednesday 5/11- Benchmark review- Unit 10
Announcements/Reminders
Agenda:
12 class DAYS until final!
Thurs 5/9- English EOC, 1st &2nd block
Mon 5/13-Benchmark #4
5/22- no school- Biology EOC
5/23- USH EOC
5/28- final exam (1st&2nd periods)
Unit 10 review
Bellwork:
Show answer
Auto Play
Slide 1 / 57
SLIDE
Similar Resources on Wayground

52 questions
WCA- Sandwiches (FRMCA)
Presentation
•
9th Grade - Professio...

54 questions
Lesson: Trends of the Periodic Table
Presentation
•
9th - 12th Grade

48 questions
Unit 4 Lesson 1 Bohr's Models and Lewis Dot Structures
Presentation
•
10th - 12th Grade

53 questions
Cardiovascular System: Heart Anatomy
Presentation
•
11th Grade - University

47 questions
Valence electrons
Presentation
•
10th - 12th Grade

51 questions
Lesson1: Motion
Presentation
•
9th - 12th Grade

51 questions
Marine Science Review
Presentation
•
9th - 12th Grade

50 questions
Lesson 1 - Taking Responsibility for Health
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade