

Thermochemistry
Presentation
•
Chemistry
•
10th Grade
•
Medium
+3
Standards-aligned
Mihir Paranjape
Used 24+ times
FREE Resource
9 Slides • 18 Questions
1
Thermochemistry
By Mihir Paranjape
2
Heat
Heat is Thermal Energy which is transferred due to a difference in Temperature.
Heat is transferred from higher temperature to lower temperature.
Heat is transferred by collissions between molecules. At higher temperature, molecules have more Kinetic Energy (as they move faster), these collide with molecules with lower Energy thereby transferring this energy.
Units are Joules (J) or Calories (cal)
Some text here about the topic of discussion
3
Temperature
Temperature is the average Kinetic Energy of molecules within a system.
Difference in Temperature causes Heat to Flow.
Can be measured using a Thermometer.
Units - C, F, K, R
K = C+273
Some text here about the topic of discussion
4
Exothermic Processes
Heat EXITS the system or flows out.
Given a negative sign. Imaging Money flowing out of your account which is bad and hence negative.
This heat goes to the surroundings, which feel warmer.
As heat leaves the system, molecules SLOW DOWN.
Gas > Liquid > Solid is example of Exothermic Process as Molecules are slowing down.
Some text here about the topic of discussion


5
Endothermic Processes
Heat ENDERS the system or flows in.
Given a positive sign. Imaging Money flowing in your account which is good, will increase your balance and hence positive.
This heat enters from surroundings, which feel colder.
As heat enters the system, molecules SPEED UP.
Solid > Liquid > Gas is example of Endothermic Process as Molecules are speeding up.
Some text here about the topic of discussion


6
Multiple Choice
During an exothermic reaction, heat content in the surroundings increases because
heat energy is destroyed during reactions
the reaction absorbs heat energy
the energy contained in the reactants is lower then the products
the energy contained in the products is lower than the reactants
7
Multiple Choice
8
Phase Changes
Some text here about the topic of discussion

9
Multiple Choice
What is the phase change of a solid to a liquid?
freezing
melting
boiling
condensation
10
Multiple Choice
11
Multiple Choice
What happens to particles when energy is added (heated)?
They speed up and spread out
They slow down and compress
They stop moving
They move closer together and speed up
12
Multiple Choice

13
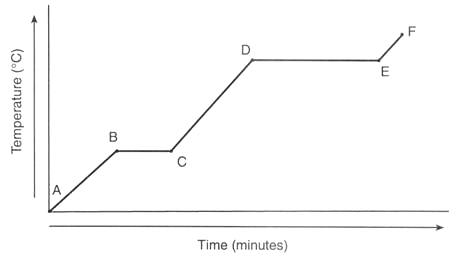
Heating Curve
Some text here about the topic of discussion

Start with Ice (SOLID) and heat it.
Ice will rise in Temp till it reaches 0C.
It will then melt to water (LIQUID). Temp does not change.
Water(LIQUID) will then heat up as more heat is added.
Water(LIQUID) will rise in Temp to 100C
At 100C, Water changes to Steam.(GAS)
14
Multiple Choice

15
Multiple Choice
16
Multiple Choice

17
Multiple Choice

18
Heating Curve
Some text here about the topic of discussion

Slope Part: (Red)
Temp increases as Heat is added.
Q = m x c x ΔT
Q = Heat (J)
m = mass (g)
c = Specific Heat (J/gC)
ΔT = Change in Temp (Tf-Ti)
19
Multiple Choice
20
Multiple Choice
How many joules of heat are needed to raise the temperature of 10.0 g of aluminum from 22.0°C to 55.0°C, if the specific heat of aluminum is 0.903 J/g°C?
298 Joules
0.003 Joules
297 J/g°C
0.003 J/g°C
21
Multiple Choice
The specific heat of aluminum is 0.9025 J/g°C. How much heat(Q) is released when a 10.0 g piece of aluminum foil is taken out of the oven and cools from 100.0° to 50.0°?
451 J
45.1 J
400 J
22
Multiple Choice
20.0 g of water. specific heat of water is 4.184 J/g°C. temperature changes from 25.0° C to 20.0° C, how much heat energy (Q) moves from the water to the surroundings?
418 Joules
209 J
83 J
4.18 J
23
Heating Curve
Some text here about the topic of discussion

Flat Part: (Blue)
Temp remains same as Heat is added.
All Heat is going towards Phase Change, called latent (lost) heat.
Q = m x ΔH
Q = Heat (J)
m = mass (g)
ΔH = Latent Heat (J/g)
24
Multiple Choice
Latent heat is
the heat absorbed or released at a boiling process during a change of phase
the heat absorbed or released at a constant temperature during a change of phase
the heat absorbed or released at a constant temperature during a change of phase of 1 kg object
the heat absorbed or released by an object to increase or decrease its temperature by 1 degree Celcius
25
Multiple Choice

How much energy is required to freeze 150 g of water? [lv=2260000 J kg−1; lf=334000 J kg−1]
50 100 cal
339 000 J
339 000 cal
50 100 J
26
Multiple Choice
How much energy is required to change 5 kg of ice at 0 ° C into water at the same temperature? (latent heat of fusion is 3.36 X 106 J/kg )
16.1 XundefinedJ
16.8 XundefinedJ
15.1 Xundefinedcal
16.8 Xundefinedcal
27
Multiple Choice
How much energy is required to melt down a 2000 g of mercury?(latent heat of fusion is 1.14 x 104 J/kg)
2.55 x undefinedJ
2.28 x undefinedJ
2,280 x undefinedJ
3.28 x undefinedJ
Thermochemistry
By Mihir Paranjape
Show answer
Auto Play
Slide 1 / 27
SLIDE
Similar Resources on Wayground

20 questions
Commas with dep and ind clauses
Presentation
•
10th Grade

21 questions
Best Method: Solving Systems of Equations
Presentation
•
7th - 10th Grade

21 questions
Cinco De Mayo
Presentation
•
10th Grade

21 questions
ADVERBIOS
Presentation
•
10th Grade

21 questions
Introduction to Trigonometry
Presentation
•
10th Grade

21 questions
Cold War Intro
Presentation
•
KG

22 questions
Thermochemistry & Nuclear Chemistry Review
Presentation
•
10th Grade

20 questions
Periodic Families
Presentation
•
9th - 10th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade