

VERPID LESSON
Presentation
•
Science
•
8th Grade
•
Medium
Standards-aligned
Desiree Kilpatrick
Used 8+ times
FREE Resource
6 Slides • 19 Questions
1
VERPID
valence electrons: Reactivity
protons: Identity
Valence Electrons and Protons
2
Multiple Choice

3
Multiple Choice

4
Multiple Choice
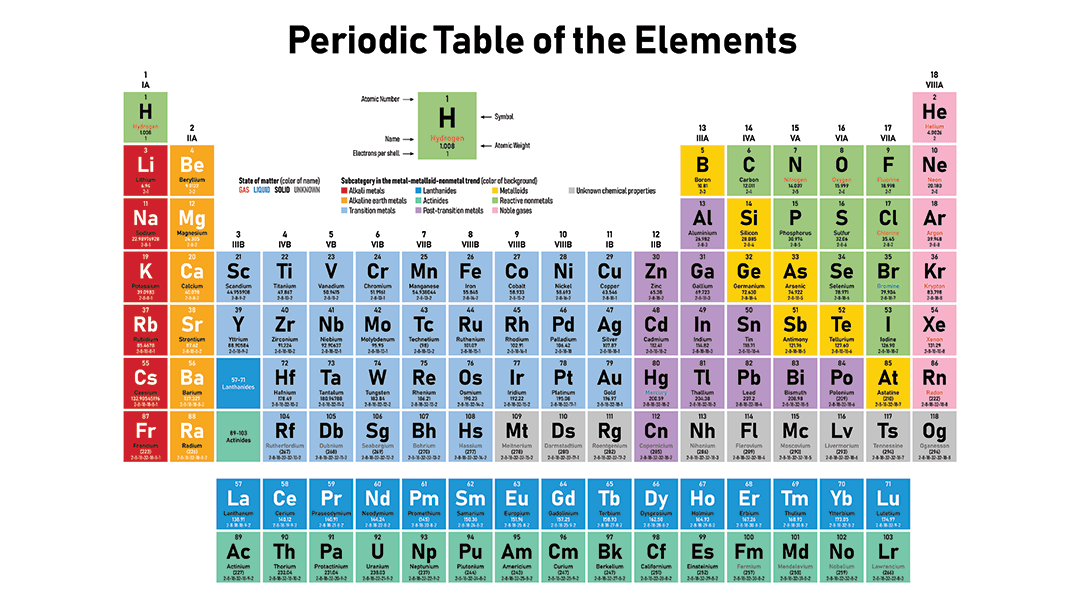
Chlorine ( Cl) has _________ valence electrons.
3
5
7
8
5

valence elctrons determne reactivity
Anything to do with reactivity, bonding, or reacting is caused/determined by Valence electrons.
6
An element wants to be stable
Stable means a full outer shell of 8 electrons (except Helium which is "full" with only 2)
Elements will add or lose electrons to have a full outershell
Subject | Subject
Some text here about the topic of discussion
7
Multiple Choice
What are valence electrons?
The total number of electrons in an atom
The number of electrons in the outermost shell
The number of electrons in the second shell
The number of protons in the outermost shell
8
Multiple Choice
All atoms are most stable with (or would "prefer") how many electrons in their valence shell?
1
2
8
18
9
Multiple Choice
When trying to identify an unknown element, a scientist determines what other elements reacts with chemically. Which property of the unknown element determines the other element it reacts with?
The total number of neutrons in the unknown element
The total number of particles in the nucleus of the unknown element
The number of protons in the nucleus of the unknown element
The number of valence electrons in the unknown element
10
Multiple Choice

11
Protons determine the identity of an element.
Each element has a unique number of protons and that number never changes for that element.
Protons: Identify
VERPID

12
Multiple Choice

Elements can have the same number of protons
13
Multiple Choice
An atom with atomic number 6 would have how many protons?
6
12
3
14
Multiple Choice

What element is represented in this Bohr Model?
Carbon
Hydrogen
Aluminum
Lithium
15
Multiple Choice

16
Multiple Choice

17
Multiple Choice
What element contains 7 protons and 5 valence electrons?
Boron
Vanadium
Nitrogen
Lithium
18
Multiple Choice

Four students were asked to name the parts of an atom that determine the atom’s identity and chemical properties. The students’ responses are shown in the table below.
Which student’s responses are correct?
Student 1
Student 2
Student 3
Student 4
19
Fill in the Blanks

Type answer...
20

21

22
Multiple Choice
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
23
Multiple Choice
24
Multiple Choice
Elements in a .................. have similar chemical properties.
period
group
row
25
Multiple Choice
Find an element with similar chemical properties to Barium (Ba).
Ca an Ra, because they are in the same group and have similar chemical properties.
Cs and La, b/c they are in the same period number.
Ca and Y, b/c they are 90 degree angle and have similiar properties.
VERPID
valence electrons: Reactivity
protons: Identity
Valence Electrons and Protons
Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

21 questions
Seasons
Presentation
•
8th Grade

21 questions
SSA Review
Presentation
•
8th Grade

17 questions
Elements, Compounds, Molecules, oh my!
Presentation
•
8th Grade

19 questions
Behavior of Waves
Presentation
•
7th - 8th Grade

19 questions
Nutrition
Presentation
•
8th Grade

18 questions
Newton's Laws of Motion Examples
Presentation
•
8th Grade

18 questions
Fossils and Geologic Time Review
Presentation
•
8th Grade

18 questions
Newton's 3rd Law of Motion
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade