

LL U5 Advanced Stoich
Presentation
•
Science
•
12th Grade
•
Easy
Standards-aligned
Jolene Kayser
Used 6+ times
FREE Resource
4 Slides • 8 Questions
1
Advanced Stoichiometry
Take notes on each of the 3 videos
College Chem Unit 5
2
Multiple Choice
the units for molar mass are...
g/mol
mol/mol
g
mol
3
Multiple Choice
What is the correct set up to find how many liters
of Oxygen are in 41.00 g of Oxygen?
141.00 g O2×32.00 g O21 mol O2×1 mol O26.02 x1023 L O2
141.00 g O2×16.00 g O21 mol O2×1 mol O26.02 x1023 L O2
141.00 g O2×32.00 g O21 mol O2×1 mol O222.4 L O2
141.00 g O2×16.00 g O21 mol O2×1 mol O222.4 L O2
4
5
Reorder
2 C8H18(l) + 25 O2(g) ----> 16 CO2(g) + 18 H2O(g) is the reaction between gasoline (octane) and oxygen that occurs inside automobile engines. If 4.00 moles of gasoline are burned, what volume of oxygen is needed if the pressure is 0.953 atm, and the temperature is 35.0°C?
Reorder the following
1Mole
Mole Ratio
Ideal Gas Law
Done
6
Drag and Drop
How many grams of water would be produced if 20.0 liters of oxygen were burned at a temperature of -10.0°C and a pressure of 1.3 atm? What is the set up
7
Multiple Choice
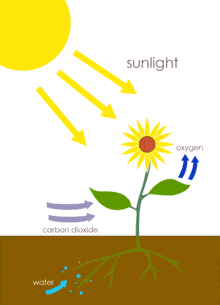
Is photosynthesis an Exothermic or an Endothermic reaction?
Endothermic
Exothermic
8
9
Multiple Choice
solute
solvent
10
Multiple Choice
Which of the following is the correct formula for calculating molarity?
moles/liters of solution
moles/kg of solvent
# particles/Avogadro's #
theoretical yield/actual yield
11
12
Drag and Drop
0.025L of 0.350 M NaOH are added to copper (II) sulfate. How many grams of copper (II) hydroxide will precipitate?
2 NaOH(aq) + CuSO4(aq) --> Cu(OH)2(s) + Na2SO4(aq)
Advanced Stoichiometry
Take notes on each of the 3 videos
College Chem Unit 5
Show answer
Auto Play
Slide 1 / 12
SLIDE
Similar Resources on Wayground

10 questions
Factors affecting the products of electrolysis
Presentation
•
12th Grade

10 questions
Cardiovascular System
Presentation
•
University

9 questions
Rivers and Streams Adaptations Notes
Presentation
•
12th Grade

10 questions
Meet and Greet your Teacher
Presentation
•
12th Grade

9 questions
Brain Scans
Presentation
•
12th Grade

8 questions
Gay-Lussac Law
Presentation
•
12th Grade

8 questions
science
Presentation
•
KG

9 questions
The Common Ion Effect
Presentation
•
12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Science

20 questions
Earth Day Trivia
Quiz
•
9th - 12th Grade

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

20 questions
Earth Day
Quiz
•
3rd - 12th Grade

20 questions
Cladograms and Phylogenetic Trees
Quiz
•
9th - 12th Grade

31 questions
STAAR Strand 1 TEKS(11,12)
Quiz
•
9th - 12th Grade

16 questions
Taxonomy and Classification
Quiz
•
6th - 12th Grade

50 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

37 questions
Hydrosphere CSA wayground review
Quiz
•
9th - 12th Grade