

Benchmark Review: States of Matter and Phase Changes
Presentation
•
Chemistry
•
8th Grade
•
Medium
Standards-aligned
Stephanie Artis
Used 41+ times
FREE Resource
15 Slides • 13 Questions
1

Benchmark Review, 2022
States of Matter &
Phase Changes
2

3
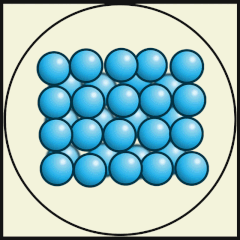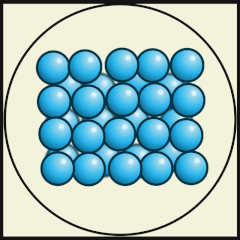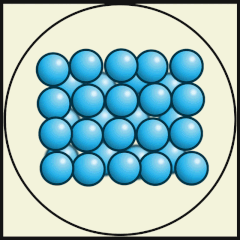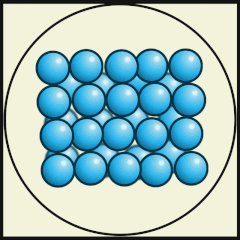
Solid
In a solid, particles are packed tightly together so they don't move much, but are dense. The electrons of each particle are constantly in motion, giving the atoms a small vibration, but they are fixed in their position.
Solids have a definite shape, as well as mass and volume, and do not conform to the shape of the container in which they are placed.
4
Liquid
In a liquid, the particles are more loosely packed than in a solid, but closely enough that it is not easily compressible.
The particles are able to flow around each other, giving the liquid an indefinite shape. Therefore, the liquid will conform to the shape of its container.

5
Gas
In a gas, the particles have a great deal of space between them, so they move easily, and have high kinetic energy (energy of movement).

6
Gas
A gas has no definite shape or volume. If unconfined, the particles of a gas will spread out indefinitely (forever). However, if confined, the gas will expand to fill its container.

7
Multiple Choice
A substance with particles that vibrate but stay in the same position is a ___.
solid
liquid
gas
plasma
8
Multiple Choice
Of the 3 states listed below, which has the fasted moving particles?
solid
liquid
gas
9
Multiple Choice
In which state of matter do the particles move the slowest?
solid
liquid
gas
10
Multiple Choice
The particles are closest together in a ___________.
solid
liquid
gas
plasma
11
Multiple Choice
solid
liquid
gas
solid, liquid and gas
12
Multiple Choice
solid
liquid
gas
solid, liquid and gas
13
Multiple Choice

The diagram to the left represents particles in a gas. (The length of the arrow represent the amount of particle energy.) Which of the diagrams below best represents the particles after the gas is heated?
14
Phase Changes
Phase changes are changes in the state of matter.
For example: when a solid melts and becomes a liquid, that is a phase change.

15
What causes a phase change?
The single most important factor involving a phase change is heat.

16

Recall that a phase change graph is used to show temperature change in a substance as it absorbs or releases heat energy.
Phase Change Graphs
17
18
Reminders...
The segments that are diagonal
( / ) are where the temperature is changing = state of matter
-Horizontal (flat) segments are where heat energy is still being absorbed BUT temperature is no longer changing = a phase change occurring

19
Multiple Choice

In this phase change graph, which line segments represent states of matter?
A, C, and E
B and D
20
Multiple Choice

On the phase change graph, which letter represents the substance as a gas?
A
B
C
D
E
21

We know that that states of matter are the slanted lines because that is where the temperature is changing.
The state of matter that has the highest temperature has to be the gas.
Therefore, the letter E represents the substance as a gas.
Where's the Gas?
22
Multiple Select

Where on the graph are phase changes taking place?
between points A and B
between points B and C
between points C and D
between points D and E
23

Recall that as a substance is absorbing heat, the only time the temperature will stop changing is when a phase change is occurring.
The only segments where the temperature is not changing is between B & C and between D & E.
Phase Changes = no change in temp
24
Multiple Choice

Which segment indicates a gas increasing in temperature?
A
B
C
D
E
25
Multiple Choice

If the substance is being cooled, what change of state (phase change) is represented by line segment B?
melting
freezing
boiling
condensing
26

If the substance is being cooled, then the temperature is decreasing, which means we have to read the graph from right to left. (from high temp to low temp)
Therefore, the phase changes will be exothermic, which are the cooling phase changes.
If we know that C is a liquid and A is a solid, then B is the phase change that occurs from liquid to solid- which is FREEZING.
Exothermic phase changes:
(Removing heat energy)
27
Multiple Choice

If the substance is being cooled, what change of state (phase change) is represented by line segment D?
melting
freezing
boiling
condensing
28

If the substance is being cooled, then the temperature is decreasing, which means we have to read the graph from right to left. (from high temp to low temp)
Therefore, the phase changes will be exothermic, which are the cooling phase changes.
If we know that E is a gas and C is a liquid, then D represents the phase change that occurs from gas to liquid- which is condensation.
Exothermic phase changes:
(Removing heat energy)

Benchmark Review, 2022
States of Matter &
Phase Changes
Show answer
Auto Play
Slide 1 / 28
SLIDE
Similar Resources on Wayground

23 questions
Atoms, History & Basic Structure - Mr. Stange (2020)
Presentation
•
8th Grade

21 questions
Best Method: Solving Systems of Equations
Presentation
•
7th - 10th Grade

21 questions
Earth Layers
Presentation
•
8th Grade

21 questions
Two Way Table Introduction
Presentation
•
8th Grade

21 questions
Cold War Intro
Presentation
•
KG

20 questions
Atomic Theory
Presentation
•
8th Grade

24 questions
Magnetic Fields
Presentation
•
8th Grade

18 questions
Lesson 5.5: The Theory of Plate Tectonics
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

36 questions
6th Grade Math STAAR Review
Quiz
•
6th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

10 questions
Exploring Properties and Models of Acids and Bases
Interactive video
•
6th - 10th Grade

20 questions
Counting Atoms Practice
Quiz
•
8th Grade

41 questions
Atomic Structure and Periodic Table Unit Review
Quiz
•
8th Grade

15 questions
Acids and Bases Review
Quiz
•
8th Grade

20 questions
Chemical Reactions
Quiz
•
8th Grade

14 questions
Mastering Balancing Chemical Equations
Interactive video
•
6th - 10th Grade

10 questions
Exploring Solutions and Their Properties
Interactive video
•
6th - 10th Grade

20 questions
Heating and Cooling Curves
Quiz
•
8th Grade