

Elements and Compounds
Presentation
•
Chemistry
•
8th Grade
•
Medium
Michael Spowart
Used 8+ times
FREE Resource
10 Slides • 20 Questions
1

Learning Outcomes:
State the names and symbols of the first
twenty elements
Describe what a mixture, molecule and a
compound is
Write word and symbol equations to show
the formation of compounds
Title Atoms, elements and compounds
2







An element is a substance that is made of only one type of atom.
Elements are the simplest substances in the universe.
Copper is an
element made up
of copper atoms
only.
Carbon is an
element made up
of carbon atoms
only.
Helium is an
element made
up of helium
atoms only.
Elements:
3



Molecules are made up of two or more atoms. Molecules can be formed
by two atoms of the same element, or by atoms from two or more
different elements.
Oxygen is an element made up
of oxygen atoms only.
How many atoms are there in
one oxygen molecule?
Other elements that form
molecules include hydrogen,
nitrogen, fluorine, iodine,
chlorine and bromine.
These are diatomic elements.
Molecules:
4
Multiple Choice
Elements are made up of only 1 type of atom
true
false
5
Multiple Choice

Water is a compound.
True
False
6






Write down whether you think each diagram shows an
element or a non-element.
Extension task (ET) Describe the particles in an element
and a non-element.
1
5
4
3
2
Activity 1:
7

1. Non – element
2.Element
3.Element
4.Non-element
5.Non-element
Stretch: The particles in an element are all the
same (1) but the particles in a non-element are
different(1).
0-2
3-5
6-7
0-1
2-3
4-5
With
stretch
Progress Check:
GREEN PEN
8



When two or more elements react in a chemical reaction,
the atoms do not just mix together, they become
chemically bonded to one another to make compounds.
Compounds can only be separated into the elements they
were made from using chemical reactions.
hydrogen
oxygen
water
+
+
Compounds:
9



10
Multiple Choice

This picture represents which of the following?
element
compound
11
Multiple Choice

This picture represents which of the following?
element
compound
12
Multiple Choice

This picture represents which of the following?
element
compound
13
Multiple Choice
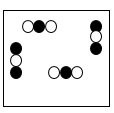
A mixture of two elements
A pure compound
A mixture of two compounds
A mixture of a compound & element
14
Multiple Choice
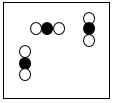
A mixture of two elements
A pure compound
A mixture of two compounds
A mixture of a compound & element
15
Multiple Choice

This picture represents which of the following?
element
compound
16

Each element can be represented by a symbol.
For many elements, the symbol is the start of the name, for
example H = hydrogen or Li = lithium. Can you think of any other
symbols like this?
However, some of the symbols are not always as you might expect;
for example, Pb = lead. Can you think of any other elements with
unexpected symbols?
The first letter of an element’s symbol is always a capital letter,
e.g. N (not n) for nitrogen.
If there are two letters in the element’s symbol, the second letter
is always a small letter, e.g. Co (not CO) for cobalt.
Element Symbols:
17
Multiple Choice
Which is NOT an element?
Co
H2
S
NaCl
18
Multiple Choice
Which is NOT a compound?
CO2
H2O
S
SiO2
19

Li2O
CO2
H2O
SO4
Fe2O4
Ba2Cu3O7
H2SO4
Li2HPO4
C6H12O6
We can tell how many elements there are by
counting the number of capital letters. All elements
start with a capital.
Na2PFO3
20

Li2O = 2
H2O = 2
SO4 = 2
Fe2O4 = 2
MgCl2 = 3
H2SO4 = 3
C6H12O6 = 3
Ba2Cu3O7 = 3
Li2HPO4 = 4
Na2PFO3 = 4
21
Multiple Choice
Is Iron (Fe) an element or a compound?
element
compound
22
Multiple Choice

Element
Compound
23
Multiple Choice
Is sugar (C12H22O11) an element or a compound?
element
compound
24
Multiple Choice
How many elements are in C12H22O11
3
11
12
22
45
25
Multiple Choice
Is salt (NaCI) a compound or a element
Element
Compound
26
Multiple Choice
Which of the following represents an element?
H2O
H2
NaCl
CaCO3
27
Multiple Choice
How many elements are represented in the compound?
Na2CO3
1
2
3
6
28
Multiple Choice
What is H2SO4
made up of?
2 hydrogen, 1 sulfur, 4 oxygen
1 hydrogen, 2 sulfur, 4 oxygen
2 hydrogen & 4 sodium
1 hydrogen & 6 sodium
29
Multiple Choice
made up of?
2 hydrogen, 1 sulfur, 4 oxygen
1 hydrogen, 2 sulfur, 4 oxygen
2 hydrogen & 4 sodium
1 hydrogen & 6 sodium
30
Multiple Choice
Na2CO3
1
2
3
4

Learning Outcomes:
State the names and symbols of the first
twenty elements
Describe what a mixture, molecule and a
compound is
Write word and symbol equations to show
the formation of compounds
Title Atoms, elements and compounds
Show answer
Auto Play
Slide 1 / 30
SLIDE
Similar Resources on Wayground

21 questions
Parts of the atom
Presentation
•
7th - 8th Grade

23 questions
Product and Quotient of Powers
Presentation
•
8th Grade

23 questions
Volume of Spheres
Presentation
•
8th Grade

23 questions
Introduction to Systems of Equations Word Problems
Presentation
•
8th Grade

23 questions
Chemical Changes
Presentation
•
8th Grade

23 questions
Physical Properties
Presentation
•
8th Grade

23 questions
WW2
Presentation
•
8th Grade

20 questions
Atoms
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

15 questions
Pythagorean Theorem Word Problems Quizizz
Quiz
•
8th Grade

20 questions
Graphing Inequalities on a Number Line
Quiz
•
6th - 9th Grade

4 questions
Spring Break rest and recharge
Presentation
•
6th - 8th Grade

20 questions
Scatter Plots and Line of Best Fit
Quiz
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade