

Atoms, Tek 8.5B, and 8.5C
Presentation
•
Science
•
•
Practice Problem
•
Medium
+2
Standards-aligned
Alex Davila
Used 15+ times
FREE Resource
18 Slides • 28 Questions
1
Atoms
Review

2
Poll
How comfortable are you with understanding what atoms are made of and how the atom is structured?
VERY COMFORTABLE!!!
Eh, pretty comfortable
I get it a little...
What are atoms?
3
What are atoms made of?
Atoms are made of Protons and Neutrons, located in the Nucleus, and Electrons around the outside of the nucleus.

4
Multiple Select
What particles are atoms made of? (Check ALL that apply:)
Protons
Neutrons
Electrons
Nucleus
5
Multiple Select
What particles are in the nucleus?
Protons
Neutrons
Electrons
Nucleus
6
Multiple Select
Which particle is outside the nucleus?
Protons
Neutrons
Electrons
Nucleus
7
Protons, Neutrons, and Electrons:
Protons and neutrons have mass.
Electrons don't have mass.

8
Multiple Select
Which two particles have mass?
Protons
Neutrons
Electrons
Nucleus
9
Particle Charges:
Protons are positively charged.
Neutrons are neutrally charged.
Electrons are negatively charged.

10
Multiple Choice
Protons are:
Positive
Neutral
Negative
11
Multiple Choice
Neutrons are:
Positive
Neutral
Negative
12
Multiple Choice
Electrons are:
Positive
Neutral
Negative
13
WORK ON REVIEW UNTIL YOU GET TO VALNENCE ELECTRONS
4MIN
14
Valence Electrons
Electrons on the outter shell are called "valence electrons."
The first shell can hold 2 electrons.
The 2nd shell can hold 8 electrons.
The 3rd shell can hold 8 electrons.

15
Multiple Choice

How many valence electrons in this diagram:
1
3
5
7
16
Multiple Choice

How many valence electrons in this diagram:
1
3
5
7
17
Multiple Choice

How many valence electrons in this diagram:
1
3
5
7
18
Reactivity
The noble gasses (group 18) are non-reactive.
Group 1 and 17 are the most reactive.
If the atom has 1 valence electron it is easily lost and reacts easily (group 1)
If the atom has 7 valence electrons it wants to grab 1 more and reacts easily (group 17)

19
Multiple Choice
Which atom is the most reactive?
20
Multiple Choice
Which atom is the most reactive?
21
Multiple Choice
Which atom is the least reactive?
22
Periodic Table
Groups & Periods
Metals, Metalloids, Nonmetals

23
Groups
Up and down
Group numbers tell us the number of valence electrons (Group 17 = 7 valence electrons)
Elements in the same group have similar reactivities

24
Reactive Groups
Group 1: most reactive metals (1 valence electron)
Group 17 (7A): most reactive nonmetals (7 valence electrons)
Group 18 (8A): Nobel Gases--- least reactive, most stable, inert (8 valence electrons)

25
Periods
Horizontal (side-to-side)
Period number tells us the number of energy levels (rings) in an atom

26
Multiple Choice

Of the following atomic models, which represents the most reactive atom?
A
B
C
D
27
Multiple Choice
Which two elements have the same number of energy levels?
S and Se
Si and Sb
Cu and Zn
Ar and Kr
28
Metals
Luster (shiny)
Malleable (bends)
Good conductor (moves heat & electricity)

29
Metalloids
Luster (shiny)
Brittle (breaks)
Semi-conductor (kind of moves heat & electricity)

30
Nonmetals
Dull (not shiny)
Brittle (breaks)
Insulators (does not move heat & electricity)

31

32
Multiple Choice

The periodic table is organized into groups and periods of elements. The characteristics of a certain group of elements are listed below. Which of these elements is in this group?
Lithium
Strontium
Aluminum
Silicon
33
Multiple Choice
Groups run:
up and down
left to right
34
Multiple Choice
Periods run:
up and down
left to right
35
Multiple Choice

Which two elements share common characteristics based on the periodic table?
Sodium and Magnesium
Chlorine and Sulfur
Argon and Neon
Calcium and Bromine
36
Multiple Choice

How are the 3 elements in the picture organized on the periodic table?
Niobium and cobalt have the same energy levels.
Cobalt and scandium have the same energy levels.
Cobalt and scandium have the same characteristics.
Niobium and cobalt have the same characteristics.
37
Multiple Choice
Metals are on the:
Left side
Right Side
Stairstep
38
Multiple Choice
Nonmetals are on the:
Left side
Right Side
Stairstep
39

40
Multiple Choice
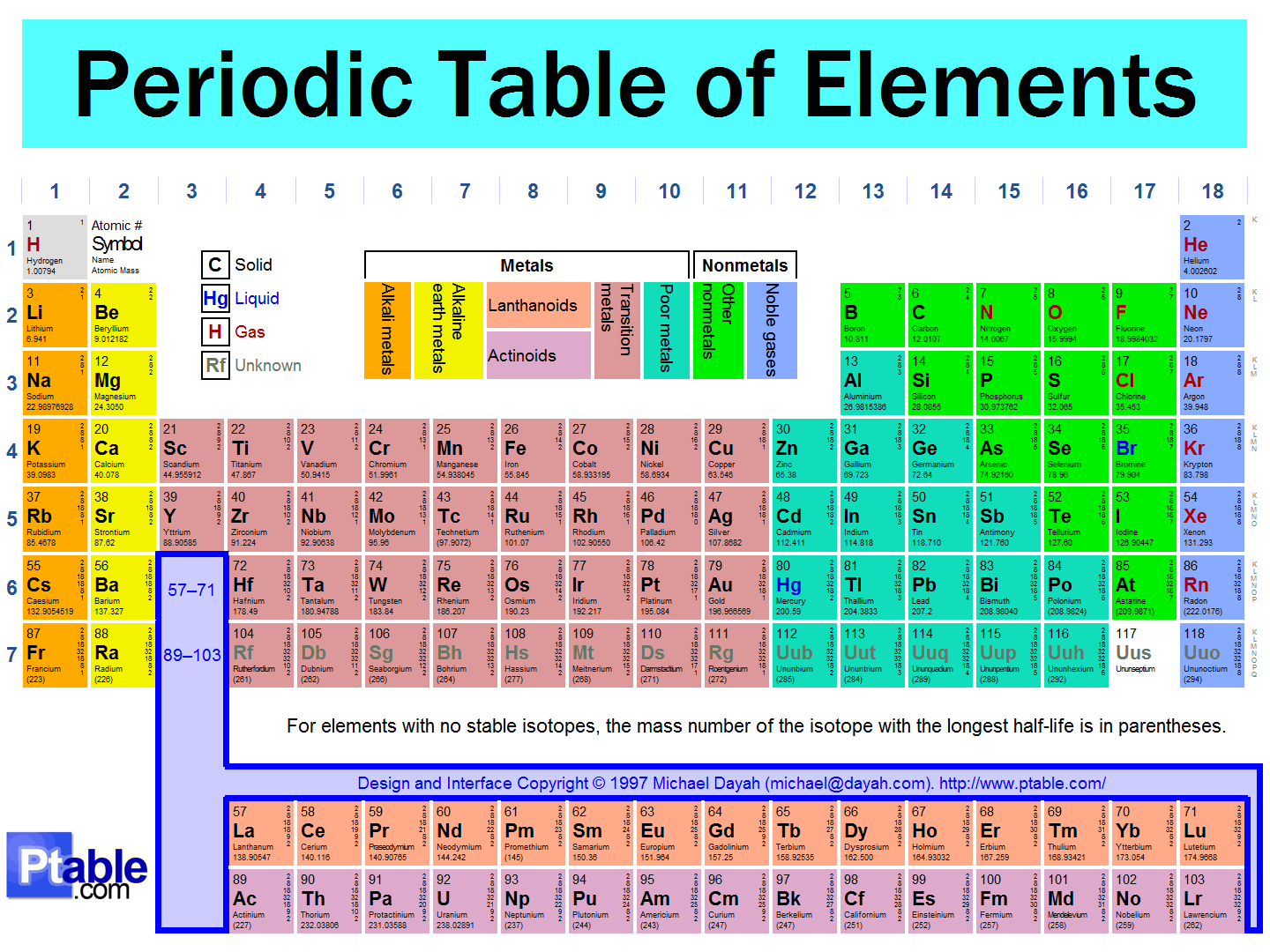
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
41

42
Multiple Choice

How many valence electrons are found in atoms of group 1?
7
6
4
1
43
Multiple Choice

USE THE PERIODIC TABLE
How many valence electrons does Phosphorus have?
31
5
15
4
44
Multiple Choice
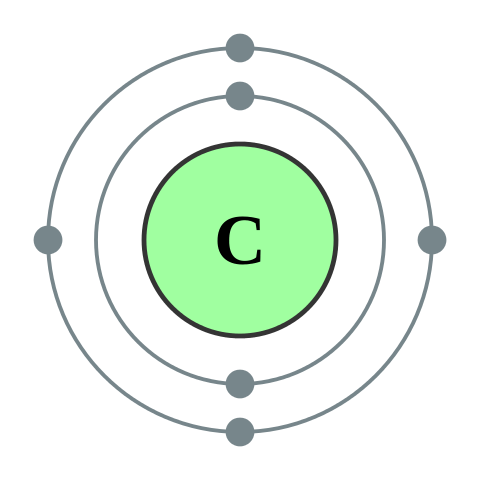
How many valence electrons does carbon have?
4
5
6
7
45
REMEMBER
VALENCE ELECTRONS - Determine REACTIVITY for elements
PROTONS- Determine the IDENTITY of ELEMENT
46
Match
Protons
Valence Electrons
Neutrons
Determine Identity of element
Determine reactivity of element
NO CHARGE,
M-A=N
Determine Identity of element
Determine reactivity of element
NO CHARGE,
M-A=N
Atoms
Review

Show answer
Auto Play
Slide 1 / 46
SLIDE
Similar Resources on Wayground

38 questions
Atoms and the Periodic Table
Presentation
•
6th - 9th Grade

38 questions
Work & Power
Presentation
•
8th - 11th Grade

42 questions
Lab Equipment
Presentation
•
8th - 12th Grade

42 questions
Metamorphic Rocks Lesson
Presentation
•
8th Grade

39 questions
PURE SUBSTANCES AND MIXTURES
Presentation
•
7th Grade

39 questions
Waves 6.P.1.1
Presentation
•
6th Grade

43 questions
light waves
Presentation
•
7th - 9th Grade

43 questions
The Cell System
Presentation
•
7th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade