

Lesson: Radioactivity
Presentation
•
Chemistry
•
11th Grade
•
Medium
Standards-aligned
William Jared Lovering
Used 36+ times
FREE Resource
22 Slides • 20 Questions
1
LESSON: RADIOACTIVITY
What is radioactivity?

2
3

4

5
Multiple Choice

How many neutrons does the isotope of lithium have?
3
4
5
8
6
Nuclear decay
is a process that occurs when an unstable atomic nucleus changes into another more stabel nucleus by emitting radiation.

7
8
Alpha Decay
An alpha particle is made of two protons and two neutrons
when an atom releases an alpha particle, its atomic number decreases by two.
Uranium-238 decays to thorium-235 through the process of alpha decay

9

10

11
Beta Decay
When beta decay occurs, a neutron in an atom changes into a proton and high-energy electron called a beta particle.
In beta decay, the atomic number of an atom increases by one because it has gain a proton.

12

13

14
Gamma Decay
Gamma rays do not contain particles, but they do contain a lot of energy.
In fact, gamma rays can pass through thin sheet of lead! because gamma rays do not contain particles.
The release of gamma rays does not change one element into another element.

15

16

17

18

19

20
Multiple Choice
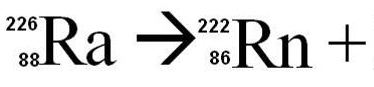
Complete the nuclear equation and determine the type of decay that is occurring in this reaction.
alpha
beta
gamma
none
21
Multiple Choice
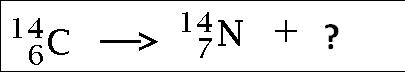
alpha particle
beta particle
gamma particle
neutron
22

23

24

25

26
Multiple Choice
What is an isotope
An atom with the same number of neutrons different number of protons
An atom that has lost or gained an electron
An atom with the same number of protons, different number of neutrons
An atom with the same number of electrons and different neutrons
27
Multiple Choice
Which particle has a negative charge
Alpha
Electron
Proton
Neutron
28
Multiple Choice
What type of decay is shown here
23892U ---> 23490Th + 42He.
alpha
beta
gamma
29
Multiple Choice
Identify the type of nuclear decay shown here
21483Bi → 0-1e + 21484Po
alpha
beta
gamma
30
Multiple Choice
remains the same
decreases by one.
increases by one.
increases by two.
31
Multiple Choice
alpha particle
beta particle
gamma ray
uranium
32
Multiple Choice
alpha, beta, gamma
X ray, beta, gamma
alpha, gamma, beta
X ray, gamma, beta
33
Multiple Choice
An alpha particle is made up of
2 protons and 4 neutrons
1 proton and 1 neutron
2 protons and 2 neutrons
4 protons and 2 neutrons
34
Multiple Choice
When a substance undergoes beta decay
The atomic number increases by 1
The atomic number decreases by 2
The atomic number does not change
35
Multiple Choice
elements with an atomic number above 81
an unstable nucleus
contaminated sewage
It decays over time
36
Multiple Choice

Protons, electrons
Protons, neutrons
Electrons, neutrons
37
Multiple Choice

Alpha radiation
Beta radiation
gamma radiation
38
Multiple Choice

an helium nucleus
an electron
a photon of light
39
Multiple Choice
Which charge does alpha particle have?
positive
negative
it has no charge
40
Multiple Choice
Which charge does a beta particle have?
positive
negative
it has no charge
41
Multiple Choice
Which charge does gamma radiation have?
positive
negative
it has no charge
42
Multiple Choice
alpha
beta
gamma
lambda
LESSON: RADIOACTIVITY
What is radioactivity?

Show answer
Auto Play
Slide 1 / 42
SLIDE
Similar Resources on Wayground

32 questions
Reaction Rates
Presentation
•
10th Grade

36 questions
Remediation 3.2 CFA Exponential Graphs
Presentation
•
11th Grade

35 questions
Color (light Waves)
Presentation
•
9th - 12th Grade

36 questions
Tema 12-proteínas y ADN
Presentation
•
10th Grade

39 questions
Chemistry Chapter 9 Section 1
Presentation
•
11th Grade

35 questions
Periodic Table Introduction
Presentation
•
10th Grade

36 questions
Chemistry Unit 4 Section 1: Properties of Liquids & Gases
Presentation
•
10th - 12th Grade

35 questions
History of the Atom
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade