

7.2.2-Matter and Its Changes
Presentation
•
Mathematics
•
9th Grade
•
Easy
Jessica Freeman
Used 5+ times
FREE Resource
45 Slides • 82 Questions
1
Physical and Chemical Changes
Feb. 1st-5th

2
Multiple Choice

Warm up. Look at the image, and calculate which formula contains the most atoms of Oxygen
1
2
3
4
3
Physical Changes (Term)
Are easily reversible, have no new products, and are often just a change of state of matter (Definition)
Examples include:
Solid to liquid
Liquid to gas
Gas to liquid
Liquid to solid

4
Chemical Changes (Term)
Are not easily reversed, new products are formed, reactants are used up, have evidence of heat/light/sound/fizzling occur, can produce electricity, or have a precipitate form (Definition)
Example: Wood Burning

5
Multiple Choice
Is ice melting a physical or chemical change?
Chemical
Physical
6
Multiple Choice
Is frying an egg a chemical or physical change
Chemical
Physical
7
Reactants (Term)
The substances in a chemical reaction that chemically combine
Examples would be the combination of sodium and chlorine

8
Products (Term)
The new substance made after a chemical reaction
Examples include Salt (NaCl) after sodium and chlorine combine

9
Precipitates (Term)
A solid that forms after two liquids chemically react (definition)
Examples would include the combination of milk and vinegar

10
Multiple Choice
When two hydrogens chemically combine with one oxygen would what would water be?
Reactant
Product
Precipitate
11
Evidence of a chemical change
Abbreviation to remember = City, Girls, Like, To, Order, Pizza
C: color change
G: Gas
L: Light is produced
T: Temperature change
O: odor is produced
P: Precipitate is formed

12
Multiple Choice
What does the o stand for in the acronym that we learned?
Gas
Light
Odor
Osmosis
13

14
Open Ended
Give Examples of a Physical Change
15

16
Open Ended
Give Examples of a Chemical Change
17
Open Ended

Dr. Garret Sho takes a piece of notebook paper from her desk. As a demonstration she tears the paper into 14 pieces.
After Dr. Sho tears the paper, what would you expect to happen to the mass of the torn pieces?
18

19
Draw
Draw an example of a Physical and Chemical Change
20

21
Fill in the Blanks
Type answer...
22

23
Multiple Choice

What is the Mass of CO2? Hint think about the Law of Conservation of Mass.
12.0g
6.2g
46.2g
20.0g
24
Multiple Choice
Review the information in the graph. Which of these statements is true?
The change from ice of liquid water is a chemical change.
The change from liquid water to water vapor is a physical change.
It takes the same amount of time for ice to melt as it does for liquid water to become water vapor.
The change from ice to liquid water to water vapor shows a decrease in mass due to the changes in states of matter.
25
Multiple Choice
There are characteristics that differentiate a chemical change from a physical change. Which statement describes a physical change?
Iron combines with oxygen to form rust
Lead is heated and becomes melts into a viscous liquid.
Sodium metal reacts explosively with oxygen
Calcium is added to acid and hydrogen gas is produced
26
Multiple Choice

Review the information in the graph. Which of these statements is true?
The change from ice of liquid water is a chemical change.
The change from liquid water to water vapor is a physical change.
It takes the same amount of time for ice to melt as it does for liquid water to become water vapor.
The change from ice to liquid water to water vapor shows a decrease in mass due to the changes in states of matter.
27
Poll
Did this help you today?
Yes
No
28
Physical Change Definition:
Change that does NOT result in a new substance.
NO new substance formed
CAN be easily reversed
Temporary change
29

30
Multiple Choice
No new substance is formed in a physical change.
True
False
31
Multiple Choice
32
Multiple Choice
Chemical channges are ______________ chnages.
Temporary
Permanent
33

34
Multiple Choice
35
Multiple Choice

What kind of change is breaking a window?
36
Multiple Choice

37
Multiple Choice

38
Multiple Choice
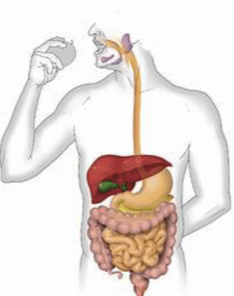
39
Multiple Choice
40
Multiple Choice

What kind of change is baking a cake?
41
Multiple Choice

What kind of change is crushing a soda can?
42
Mixtures
is a type of matter that
1 ....... is composed of 2 or more substances that are physically combined
2...... has composition that can change (vary) from one sample to another
3....... can be physically separated into its components
4....... retains the properties of the individual components
43
Examples of mixture
NaCl (aq) ........ salt water
C6H12O6 (aq) ....... sugar solution
HCl (aq) ......... Hydrochloric acid solution
soil, concrete and air are also mixture

44
Homogeneous mixture
the components of the mixture are uniformly and evenly mixed throughout
sample taken with the same mixture have the same composition

45
Heterogeneous mixtures
the components of the mixture are not uniformy mixed throughout
sample taken with the sample mixture have different composition

46
Multiple Choice
Which is a mixture of substances?
Cl2(g)
H2O(l)
MgCl2(s)
KNO3(aq)
47
Multiple Choice
Which is true of a KCl solution?
It is composed of substances that are chemically combined.
It is composed of substances that are physically combined.
It is composed of substances with the same atomic number.
It is a pure substance.
48
Multiple Choice

Which are elements?
A
B
C & D
C
49
Multiple Choice
When NaNO3(s) is dissolved in water, the resulting solution is classified as a
heterogenous compound
homogeneous compound
heterogeneous mixture
homogeneous mixture
50
Multiple Choice
One similarity between all mixtures and compounds is that both
are heterogeneous
are homogeneous
combine in definite ratio
consist of two or more substances
51
Multiple Choice
Which phase allows molecules to move around each other?
Solid
Liquid
Gas
None of the above.
52
Movement of Molecules
Solid- molecules move in place
Liquid- molecules move around each other
Gas- molecules move away from each other
53
Multiple Choice
Why does your hand feel cold as an ice cube melts on it?
Thermal energy is transferred from the ice to your hand
Thermal energy can't transfer between your hand and ice
Thermal energy is transferred from your hand to the ice
Cold energy is transferred from the ice to your hand
54

55

56
Multiple Choice

Ralph and Sheila are at a construction site and they are stacking bricks. The diagram below shows the bricks before they touch. Use the information in the diagram to answer the question. After the bricks have been touching for a while, whose top brick will be cooler?
Both will be the same temperature, because both bottom bricks transferred the same amount of energy.
Sheila's top brick was will be cooler, because the energy that transferred to Sheila's top brick was spread out with more molecules.
Ralph's top brick was cooler, because less energy has to transfer to Ralph's top brick.
57
Multiple Choice

What energy transformation occurs when a crock pot is plugged into an outlet and then the control knob is turned "on"?
thermal to electrical
mechanical to thermal
electrical to thermal
electrical to sound
58
Multiple Choice
According to the Law of Conservation of Energy, energy cannot be _________ or ____________.
destroyed, destroyed
created, saved
created, destroyed
lost, found
59
Multiple Choice

Electrical Energy is transferred to _____ Energy.
Thermal
Solar
Electrical
None
60
Multiple Choice
A hairdryer converts --
electrical energy into chemical
electrical energy into thermal
chemical energy into mechanical
thermal energy into mechanical
61
Multiple Choice
Which type of energy transformation occurs when a flashlight shines?
chemical → thermal → electrical
chemical → electrical → light
thermal → chemical → light
chemical → electrical → chemical
62
Multiple Choice
If a Lion ate a gazelle what kind of energy is being transferred?
Mechanical
Chemical
Electrical
Thermal
63
Multiple Choice
Which kind of energy change occurs when a candle is burning?
Chemical to light.
Light to electrical.
Solar to heat.
Chemical to electrical.
64
Multiple Choice

What energy transformation occurs when an alarm clock rings?
electrical to mechanical, sound
chemical to mechanical,sound
electrical to thermal, chemical
mechanical to mechanical, thermal, sound
65

Living things VS. Objects
Living things:
Eat, breathe, run, cry, get angry
Do they have energy?
Objects:
Don't eat, don't breathe, don't move, don't cry, don't get angry.
Let's remember...
66
Energy in Objects
Did you know that all objects possess energy?

67
What is energy?
It's everywhere around us and takes all sorts of forms.
How things change and move.
It takes energy to cook food, to drive to school, and to jump in the air. Different forms of Energy
"the ability to do work"

68
Multiple Choice
Choose TRUE OR FALSE: "We use energy to do everyday activities like eating, running, even sleeping"
TRUE
FALSE
69
Multiple Choice
CHOOSE TRUE OR FALSE: How we process energy is called WORK
FALSE
TRUE
70
Kinetic Energy
Pick up the bow and arrow and shoot it
Stored energy = potential energy
Transforms to moving/motion energy
Also called "KINETIC"

71
Multiple Choice

What type of energy is this?
Potential
Kinetic
72
Multiple Choice

What type of energy is it?
Potential
Kinetic
73
Multiple Choice

The water falling is using what type of energy:
Kinetic
Potential
None
All of them
74
Law of Conservation of Mass

75
Clear Learning Target:
I can define the Law of Conservation of Mass so that I can determine if examples given follow the rule.

76
Elements and Compounds
https://www.youtube.com/watch?v=i2OE9ljBKD8

77
Physical Change
A Physical Change happens when a substance changes but no new substance is formed.
Physical Change occurs when the state of a substance changes.
Freezing
Evaporating
Examples include: Water Freezing, Cutting Paper, Melting Butter, Breaking Glass

78
Chemical Change
A Chemical Change Occurs when a new substance is formed.
Indications that a new substance has formed (Chemical Change):
A change in color
Formation of Bubbles
Temperature changes
Forms a gas
Examples of a Chemical Change: Wood burning, A nail rusting, Food Rotting.

79
Multiple Choice

Is this an example of a Physical or Chemical change?
Physical
Chemical
80
Multiple Choice

Is this a image a physical or chemical change?
Physical
Chemical
81
Multiple Choice

Is this image a physical or chemical change?
Physical
Chemical
82
Law of Conservation of Mass
Antoine Lavoisier - "Revolutionized Chemistry"
Lavoisier discovered that "Mass can not be created or destroyed" by carefully measuring the product and reactants in many of the chemical reactions he was researching.
Law of Conservation of Mass states that: "Mass can not be created or destroyed"

83
Products vs. Reactants
RAP = Reactants Arrow Products





84
Products and Reactants in a Chemical Equation

85
Products and Reactants in a Chemical Reaction
Reaction between Methane and Oxygen :
All of the atoms that were present at the beginning of the reaction (reactants) are still present in the products. They have just been moved around or transformed!

86
Multiple Choice
What does the Law of Conservation of Mass state?
Mass is created at the end of a chemical reaction
Mass can not be created or destoryed
Mass is created in a physical change
New chemicals are formed when a chemical reaction takes place.
87
Counting The Atoms: Subscripts Vs. Coefficients

88
Multiple Choice

Using the picture, what is the coefficient of this chemical compound?
3
4
There isn't one
89
Multiple Choice

Using the picture, what is the subscript of this chemical compound?
4
3
There isn't one
90
Counting Atoms Review

91
Counting Atoms Review

92
Counting Atoms Review

93
Counting Atoms Review

94
Multiple Choice

What state of matter is represented in the picture above?
solid
liquid
gas
95
Multiple Choice

What state of matter is represented in this picture?
solid
liquid
gas
96
Multiple Choice
Heating a solid to a high temperature will usually result in this phase change.
Condensation
Melting
Sublimation
Evaporation
97
Multiple Choice
This state of matter expands to fill its container.
Solid
Liquid
Gas
Plasma
98
Multiple Choice
When an object has the ability to be stretched into a thin wire this is
Density
Ductility
Dirtability
Dissovility
99
Multiple Choice
The ability to hammered into a thin sheet or be bent is
Ductility
Solubility
Density
Malleability
100
Multiple Choice
101
Multiple Choice
102
Multiple Choice
103
Multiple Choice
104

105

106

107

108
Multiple Choice
Matter can not be created nor destroyed: it can only be
Destroyed a little bit
Invisible
Transformed, changed
None of the above
109
Multiple Choice
If reaction starts with 20g of reactants it should produce
a total of 40g of products
a total of 10g of products
a total of 80 g of products
a total of 20g of products
110
Multiple Choice
Mr. Brady did an experiment. The mass he started with was 15 grams. How much mass should he end with?
less than 15 grams
more than 15 grams
15 grams
111
Multiple Choice
What is the Law of Conservation of mass?
Mass is created in a chemical reaction
Mass is created in a physical change
New chemicals formed from a chemical reaction have a larger overall mass than the original reactants
Mass is never created or destroyed
112
Multiple Choice
113
Multiple Choice
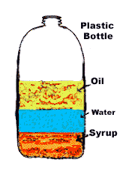
114

115
Multiple Choice
2H2 + O2 --> 2H2O
116
Multiple Choice
2H2 + O2 --> 2H2O
117
Multiple Choice

Identify the Reactants in this equation.
A
B
118
Multiple Choice
119
Multiple Choice
120
Multiple Choice
121
Multiple Choice
122
Multiple Choice
Al + O2 → 2Al2O3
123
Multiple Choice
Mg + __HCl ---> MgCl2 + H2
124
Multiple Choice
H2O
125
Multiple Choice
126
Multiple Choice
What coefficient should be used to make the following equation balanced?
N2+O2--> _NO
1
2
3
4
127
Multiple Choice
N2+O2--> _NO
Physical and Chemical Changes
Feb. 1st-5th

Show answer
Auto Play
Slide 1 / 127
SLIDE
Similar Resources on Wayground
![G7- Final Review Chapter 2 [Term1]](https://cf.quizizz.com/image/image-loader.svg)
117 questions
G7- Final Review Chapter 2 [Term1]
Presentation
•
7th Grade

121 questions
NMMS 2021 WRITING CAMP - The Conventions Pyramid!
Presentation
•
8th Grade

120 questions
Trig lesson
Presentation
•
9th Grade

117 questions
F5 Life's Great Mysteries L13 Review
Presentation
•
10th Grade

128 questions
5/6: The Early Republic & Age of Jackson PPT
Presentation
•
8th Grade

119 questions
Composite Shaded Area
Presentation
•
6th - 8th Grade

124 questions
Chemical Formulae
Presentation
•
9th - 12th Grade

119 questions
GEO Unit 1: Wind, Storms, and Cyclones
Presentation
•
6th - 8th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Mathematics

10 questions
Test Your Knowledge with 15 Fun Trivia Questions
Interactive video
•
6th - 10th Grade

21 questions
Factoring Trinomials (a=1)
Quiz
•
9th Grade

25 questions
Final Exam Review
Quiz
•
8th - 9th Grade

23 questions
Cumulative Vocabulary Practice
Quiz
•
9th - 11th Grade

25 questions
Complementary and Supplementary Angles
Quiz
•
7th - 10th Grade

21 questions
Quadratic Unit Review
Quiz
•
9th Grade

31 questions
IM1: Naming <'s, Transform. & Tri Congruence Final Review #3
Quiz
•
9th - 10th Grade

20 questions
EOC Math 1 Review
Quiz
•
9th Grade