

Chapter 9 Notes - Thermodynamics
Presentation
•
Mathematics
•
Professional Development
•
Easy
Standards-aligned
Joseph Lloyd
Used 2+ times
FREE Resource
52 Slides • 32 Questions
1
Chapter 9 - Thermodynamics

2
Open Ended
What is a calorie?
3
Open Ended
What type of energy do we burn?
4
Open Ended
So if you release heat due to chemical breakdown of molecules and movement of blood in your body, then how do you think calorie can be used in thermodynamics?
5
Open Ended
Remember from meteorology that all natural systems want to be balanced. For weather to become balanced, what would have to happen between the warm and cool air masses merging onto each other? For example, if one air masses temperature is 47 degrees F and another is 87 degrees F, and if they were merging together, how would they eventually balance out?
6
What is heat?
How is it produced?
How is it measured?
How do we use it?
Could the sun be a reliable, inexhaustible source of energy?
Chapter 9 - Thermodynamics
7
Early Theories of Heat...
Sixth century BC: Greek Philosopher Heraclitus claimed there were three natural elements—earth, water, and fire
Thirteenth century AD: philosophers and scientists thought that motion is the essence of heat
Chapter 9 - Thermodynamics

8
Early Theories of Heat...
This concept was reaffirmed by English philosopher and scientist Francis Bacon
18th century Joseph Black suggested heat was like an invisible fluid
Solids needed to be filled up with this heat-fluid until they melted
Chapter 9 - Thermodynamics

9
Early Theories of Heat...
French chemist Lavoisier developed this idea and called the heat-fluid “caloric”
The caloric theory was successful in observations and predictions
Theory of steam engines in early 1800s by Nicholas Sadi Carnot - example of thermal heat and work capacity.
Chapter 9 - Thermodynamics

10
Chapter 9 - Thermodynamics

11
Early Theories of Heat...
Carnot’s work evolved into the science of modern thermodynamics
The study of thermal energy and heat and how they relate to other kinds of energy and work
Carnot cycle is the ideal cycle, in which heat is generated, or added, at a constant temperature and rejected at a lower temperature.
Chapter 9 - Thermodynamics

12
Early Theories of Heat...
Benjamin Thompson was first to discount the caloric theory
Observed during military training cannons fired without cannonballs became much hotter than those that fired normally
Chapter 9 - Thermodynamics

13
Early Theories of Heat...
If the caloric theory were true, release of caloric and resulting temperature rise should have been unaffected by the cannonball when the gunpowder was fired
Rumsford (Thompson) observed a dull boring bit could generate more heat
Chapter 9 - Thermodynamics


14
Early Theories of Heat...
If the caloric theory were true, release of caloric and resulting temperature rise should have been unaffected by the cannonball when the gunpowder was fired
Rumsford (Thompson) observed a dull boring bit could generate more heat
Chapter 9 - Thermodynamics


15
Multiple Choice
Which scientist first challenged the caloric theory through experiments?
Antonie Lavoisier
Nicholas Sadi Carnot
Benjamin Thompson
16
Chapter 9 - Thermodynamics
Julius Robert von Mayer was first to experiment with the idea of heat as energy (not as matter) in 1842
Mayer used a horse-powered mechanism to stir a pot of paper pulp
Calculated mechanical energy needed to heat mixture
Demonstrated mechanical energy could be converted to thermal energy

17
Chapter 9 - Thermodynamics
James Prescott Joule: clear connection between mechanical energy and heat
Did experiments using mechanical devices that were dropped, shaken, and stirred to produce changes in temperature of liquids and gases

18
Chapter 9 - Thermodynamics
James Prescott Joule: clear connection between mechanical energy and heat
Did experiments using mechanical devices that were dropped, shaken, and stirred to produce changes in temperature of liquids and gases
Joule concluded that the equivalent of 4.18 N•m of mechanical work would raise the temperature of 1 g of water
1 °CEstablished that energy causes temperature changes in matter

19
Multiple Choice
Which scientist first showed a clear connection between mechanical energy and heat?
Robert Mayer
James Prescott Joule
William Thompson
20
Early Theories of Heat...
Kinetic-molecular model: atoms, molecules, ions, and subatomic particles are in constant motion and have kinetic energy
Internal energy of matter - Sum of all potential and kinetic energies in a substance
Cannot be measured
Chapter 9 - Thermodynamics

21
Multiple Choice
The sum of all potential and kinetic energies in a substance is called its
external energy
internal energy
heat
temperature
22
Early Theories of Heat...
Kinetic molecular theory states that gas particles are in constant motion and exhibit perfectly elastic collisions, meaning elastic collision is an encounter (collision) between two bodies in which the total kinetic energy of the two bodies remains the same
Example: Phet Simulation on Gases
Chapter 9 - Thermodynamics

23
Chapter 9 - Thermodynamics
Warm systems move towards cool systems due to density differences due to temperature and pressure!

24
Chapter 9 - Thermodynamics

25
Chapter 9 - Thermodynamics
High pressure "chases" Low Pressure


26
Chapter 9 - Thermodynamics

27
Chapter 9 - Thermodynamics
Why do we study this???
For a renewable energy source to be valuable, it must be able to convert its energy into a useable form
Solar panels can change sunlight directly into electricity

28
Chapter 9 - Thermodynamics
Energy costs continue to rise
Is there a more economical way of converting the free energy of the sun into electricity?
Solar thermal (ST) power plant
Mojave Desert, California

29
Open Ended
Why do you think that solar panels are still not the most beneficial resource for energy in this current time period?
30
Chapter 9 - Thermodynamics
Part 2 - How do we measure heat? Temperature
31

A measurement that describes the average kinetic energy in random thermal motion per atom or molecule
Temperature
32
Multiple Choice
33

A calibration tool to measure temperature changes
3 different scales
Fahrenheit
Celsius
Kelvin- the base unit of temperature in the International System of Units, having the unit symbol K
Thermometers
34
Multiple Choice
35
The Fahrenheit Scale
Freezing temperature of pure water
Boiling temperature of pure water
On the Fahrenheit scale there are 180 degrees between water’s freezing point, 32 °F, and boiling point, 212 °F
Thermometers

36
The Celsius Scale
Temperature range between freezing and boiling points of water: 100 degrees
Freezing: 0 °C
Boiling: 100 °C
Thermometers

37
is produced when a rise in temperature causes atoms and molecules to move faster and collide with each other
Thermal Energy

38
Multiple Choice

39
How to Convert Temperature Scales?

40
Multiple Choice

Convert 101 degrees F to degrees C
34 degrees C
38 degrees C
42 degrees C
41
Multiple Choice

Convert -38.9 degrees C to Kelvin
234.3 K
244.5 K
293.8 K
42

the energy associated with the random, disordered motion of molecules within the system
ENTROPY
the degree of disorder or randomness in the system...
Internal Energy
43

ENTROPY
the degree of disorder or randomness in the system...
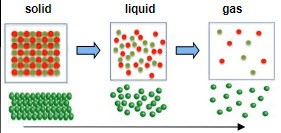
Entropy is a measure of how much the atoms in a substance are free to spread out, move around, and arrange themselves in random ways. For instance, when a substance changes from a solid to a liquid, such as ice to water, the atoms in the substance get more freedom to move around.
Internal Energy
44
Multiple Choice
45

“Heat” is often used for both thermal energy and temperature
Neither is scientifically correct
The amount of thermal energy in an object is a property of the object—it has thermal energy
Heat and Entropy
46

The more disorganized the molecules are the higher state of entropy
Entropy increases as temperature increases
Entropy is a measure depending on the disorganization
Entropy and States of Matter
47
Multiple Choice
Do molecules generally move faster at 45 C or 106 C?
45 C because molecules have more energy than in 106C
106 C because molecules have more energy than in 45C
106 C because molecules have less energy than in 45C
45C because molecules have less energy than in 106 C
48
Multiple Choice
49

All kinetic energy stops
the minimum possible
temperature, 0 K, or −273ºC (−459ºF)
Absolute Zero
50
Multiple Choice
What happens at absolute zero?
All Motions cease
everything turns to a solid
everything dies
who knows?
51

Changes both the temperature and the state of matter
Travels from high to low
Heat
52
0th Law
Thermal equilibrium
The condition under which two substances in physical contact with each other exchange no heat energy
temperatures balance out, heat stops flowing, then the system (or set of systems)
Laws of Thermodynamics

53
Multiple Choice
the energy that is transferred between objects because of a temperature difference
54
Multiple Choice

55
1st Law
Conservation of Energy
Energy cannot be created or destroyed only changed into another form
Laws of Thermodynamics

56
Multiple Choice
57
2nd Law
Heat
Heat always flows from hot to cold
Laws of Thermodynamics

58
Multiple Choice
59
Multiple Choice
60
3rd Law
Entropy
As temperature approaches absolute zero, entropy is at constant minimum
Laws of Thermodynamics

61
Multiple Choice
62
Multiple Choice
63

Conduction
the transfer of heat through or between materials by direct contact
Heat Transfer Method
64
Multiple Choice
Conduction
The transfer of thermal energy through solids
The transfer of thermal energy through liquids and gases
The transfer of thermal energy as electromagnetic waves
65

Convection
the transfer of heat by the motion of liquids and gases
Heat Transfer Method
66
Multiple Choice
Convection
The transfer of thermal energy through liquids and gases
The transfer of thermal energy through solids
The transfer of thermal energy as electromagnetic waves
67
Multiple Choice
68

Radiation
the dispersal of energy through space through electromagnetic waves
Heat Transfer Method
69
Multiple Choice
70
Multiple Choice
Radiation
The transfer of thermal energy as electromagnetic waves
The transfer of thermal energy through solids
The transfer of thermal energy through liquids and gases
71
materials that transfer heat readily (ex: diamond, copper, aluminum, gold)
Thermal Conductors

72
materials that do not transfer heat readily (ex: plastic, wool, styrofoam)
Thermal Insulators

73
Multiple Choice
74
Multiple Choice
Heat moves easily through materials called ____
Wood
Plastic
Rubber
Metal
75
Heat Capacity
Thermal energy, in joules, an object must gain or lose to cause a temperature change of 1 °C
Units: J/°C or J/K
How do we measure heat?

76
How do we measure heat?

77
How do we measure heat?

78
How do we measure heat?
Specific Heat Capacity
Specific heat (csp) is the heat capacity per gram of material—the amount of thermal energy that must be gained or lost to change the temperature of
1 g of the substance 1°C

79
How do we measure heat?

Water has an especially high heat capacity at 4.18 J/g*C, which means it takes more heat to warm a gram of water. This is why, throughout the course of a warm summer day, the water in the ocean does not experience a significant change, thus the ocean has high specific heat capacity
80
Open Ended
Describe in your own words why the ocean has high or low specific heat capacity?
81
How do we measure heat?
Water’s heat capacity allows perspiration to be an effective cooling mechanism for our bodies. Thus, when we sweat it is cooling us off!

82
How do we measure heat?
When thermal energy is added to or taken from a material, its temperature usually changes because kinetic energy of its atoms and molecules changes
Sometimes the temperature doesn’t change
83
How do we measure heat?
Latent heat of fusion (Lf): the amount of thermal energy exchanged per gram of material during melting or freezing
Latent heat of vaporization (Lv): the amount of thermal energy exchanged per gram of material during boiling or condensation
84
How do we measure heat?
Every substance has a distinctive specific heat for each of its states
Every substance has a characteristic latent heat of fusion and of vaporization
Chapter 9 - Thermodynamics

Show answer
Auto Play
Slide 1 / 84
SLIDE
Similar Resources on Wayground

74 questions
DIET Virtual Lab for Mathematics
Presentation
•
Professional Development

77 questions
Level K Sight Words
Presentation
•
KG

79 questions
Review ROBO TEST 1st December
Presentation
•
KG - University

80 questions
Level 1 C3: Verb "Be"
Presentation
•
University - Professi...

76 questions
Untitled Presentation
Presentation
•
Professional Development

78 questions
Untitled Presentation
Presentation
•
University - Professi...

81 questions
Math for College Algebra
Presentation
•
University

80 questions
INTER 3 - BUSINESS PARTNER B1
Presentation
•
Professional Development
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Mathematics

20 questions
Guess The App
Quiz
•
KG - Professional Dev...

10 questions
Food Quiz
Quiz
•
Professional Development

11 questions
NFL Football logos
Quiz
•
KG - Professional Dev...

19 questions
Minecraft
Quiz
•
6th Grade - Professio...

20 questions
Block Buster Movies
Quiz
•
10th Grade - Professi...

40 questions
Flags of the World
Quiz
•
KG - Professional Dev...

23 questions
super heros
Quiz
•
KG - Professional Dev...

11 questions
SOCCER PLAYERS AND TEAMS
Quiz
•
KG - Professional Dev...