

Biomolecules Review
Presentation
•
Biology
•
11th Grade
•
Practice Problem
•
Easy
Robert Bailey
Used 2+ times
FREE Resource
2 Slides • 33 Questions
1
Biomolecules

2
Labelling
Label the Hydrogen opposite:
Covalent Bond
Negatively charged region
Positively charged region
3
Dropdown
4
Draw
Show how water would interact with a) a positive ion
b) a negative ion
5
Drag and Drop
6
Match
Match the following monomers to their picture:
alpha- glucose
beta-glucose
amino acid
fructose
Adenosine monophosphate
alpha- glucose
beta-glucose
amino acid
fructose
Adenosine monophosphate
7
Open Ended
Explain what is meant by the terms monomer and polymer.
8
Labelling
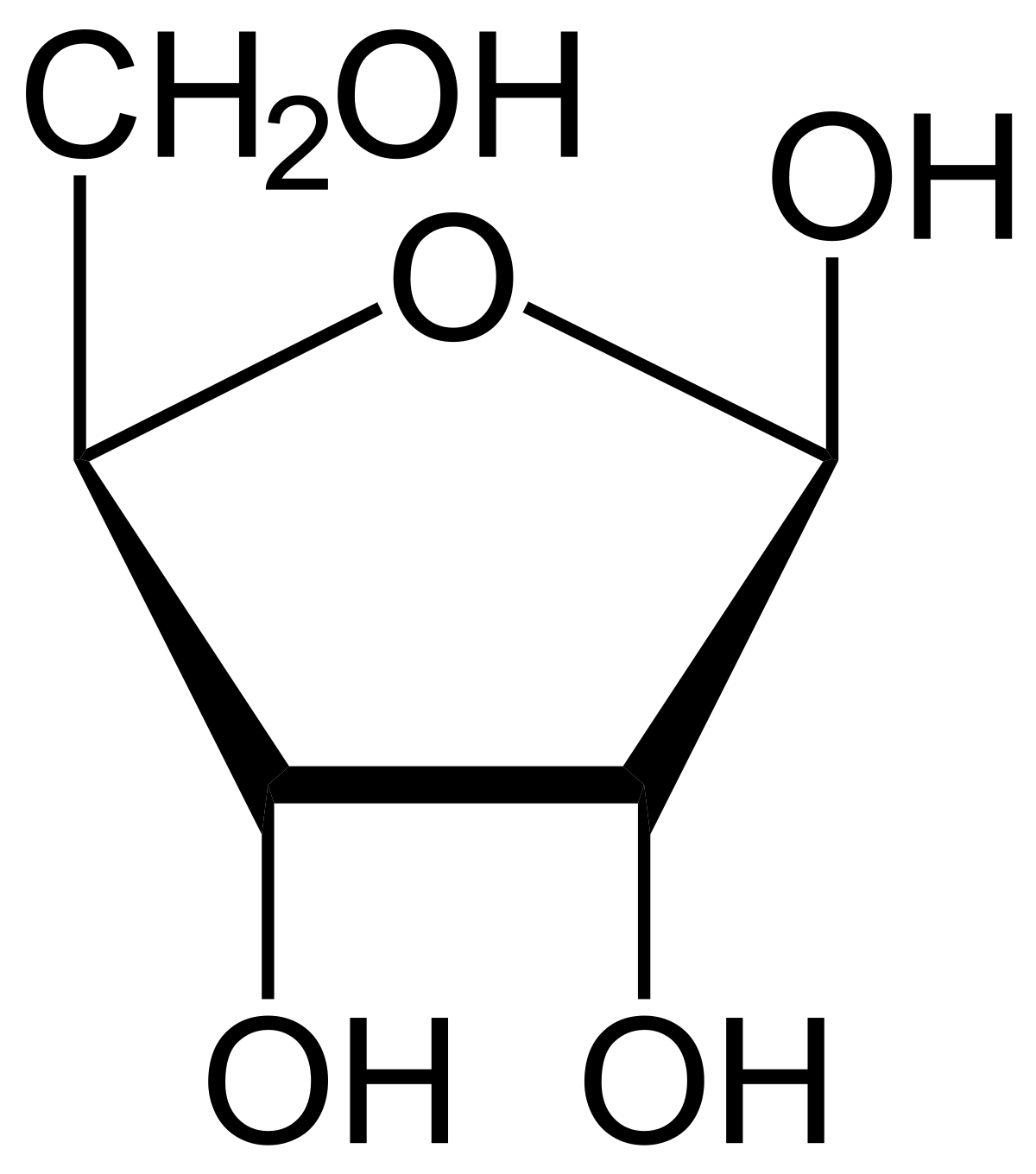
Label the diagram opposite
ribose
nitrogenous base
phosphate ion
deoxyribose
9
Drag and Drop
10
Fill in the Blanks
Type answer...
11
Match
Match the following nitrogenous bases to their complementary pair:
Adenine
Cytosine
Uracil
Thymine
Guanine
Adenine
Thymine
Guanine
Adenine
12
Categorize
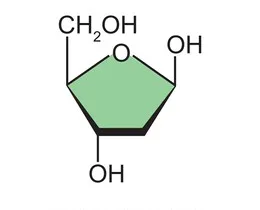
Uses the sugar ribose
Contains uracil as one of its bases
Involved in protein synthesis only
Single-stranded molecule
Found in the cytoplasm of a cell
Transcribed from DNA
Uses the sugar deoxyribose
Contains thymine as one of its bases
Stores genetic information and the code contains the code for protein synthesis
Double-stranded molecule
Found in the nucleus of a cell
Replicates to pass on genetic information
More stable than RNA
Forms a double helix structure in most organisms
Shorter molecule
Generally a longer molecule
13
Multiple Select
Which are the two purine bases?
Adenine
Thymine
Cytosine
Guanine
14
Fill in the Blanks
Type answer...
15
Hotspot
Indicate which direction is the 5' prime end of the DNA molecule
16
Open Ended
All organic molecules are carbon based. Explain why carbon is able to form large organic molecules.
17
Draw
Draw alpha-glucose
18
Categorize
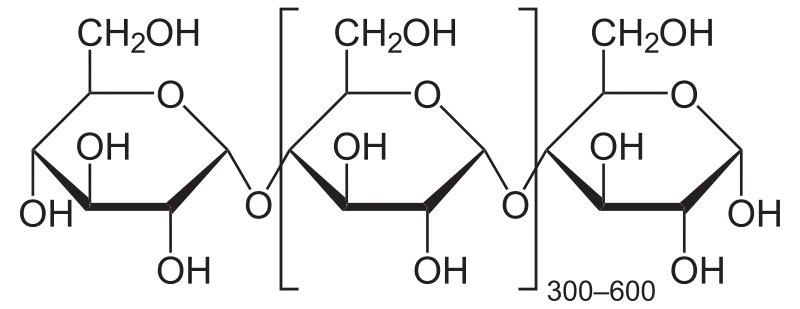
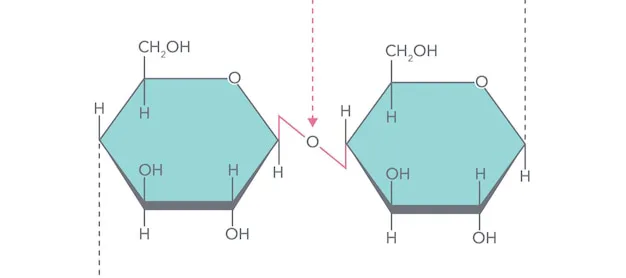
alpha-1,4-glycosidic bonds
unbranched polymer of glucose
digestible by humans
found in starch
branched polymer of glucose
contains alpha-1,4- and alpha-1,6- glycosidic bonds
introduces branches into starch
highly branched polymer of glucose
energy storage in animals
alpha-1,4-glycosidic bonds with alpha-1,6-glycosidic branches
similar structure to amylopectin
beta-1,4-glycosidic bonds
structural component of plant cell walls
indigestible by humans
linear polymer of glucose
found in plant fibers
Forms a helix
Every second glucose rotated 180 degrees
19
Match
Match the following:
Glucose + Fructose
Glucose + Galactose
Glucose + Glucose
Sucrose + Water
Lactose + water
Maltose + Water
Sucrose + Water
Lactose + water
Maltose + Water
20
Open Ended
Explain why the structure of glycogen and starch make them good for energy storage.
21
Drag and Drop
22
Labelling
Label the diagram
amino group
R group
carboxylic acid
23
Drag and Drop
24
Match
Match the following:
Primary
Secondary
Tertiary
Quaternary
Primary
Secondary
Tertiary
Quaternary
25
Categorize
Prosthetic groups
Multiple polypeptide chains
determined by tertiary structure
Ionic bonds
Disulphide bridges
determined by secondary structure
One polypeptide chain
hydrogen bonding only
alpha helix
beta- pleated sheets
Sequence of amino acids
determines secondary, tertiary and quaternary structu
Organize these options into the right categories:
26
Multiple Choice
How many naturally occurring amino acids are there
20
24
21
19
27
Dropdown
28
Drag and Drop
29
Match
Match the following:
Glycerol
Fatty acid
Phospholipid
Triglyceride
Cholesterol
Glycerol
Fatty acid
Phospholipid
Triglyceride
Cholesterol
30
Drag and Drop
31
Categorize
No C=C
One C=C
At least 2 C=C
typically found in animals
Often solid
Can pack tightly
At least one bend
Packs less tightly
Large kinks in tail
Packs loosely
Typically Liquid at room temperature
Often found in plants
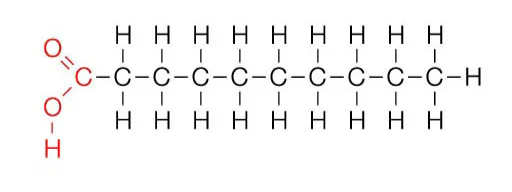
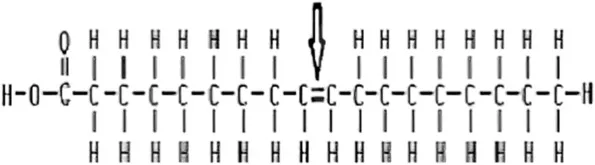
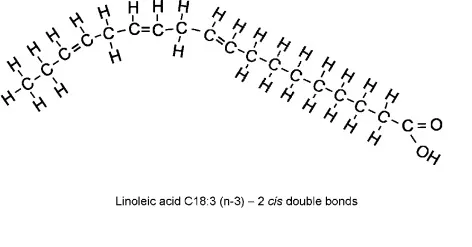
Organize these options into the right categories:
32
Drag and Drop
33
Fill in the Blanks
Type answer...
34
Open Ended
How are fats suited to their function as a long term energy storage molecule?
35
That's all folks!
Biomolecules

Show answer
Auto Play
Slide 1 / 35
SLIDE
Similar Resources on Wayground

30 questions
Introducción al Curso de Computación - Nivel B
Presentation
•
KG

28 questions
EARTH SCIENCE REVIEW
Presentation
•
11th Grade

28 questions
Насекомые - вредители растений
Presentation
•
KG

32 questions
Reproduction of Representative Animals
Presentation
•
11th - 12th Grade

30 questions
SISTEM PENCERNAAN PADA MANUSIA
Presentation
•
11th Grade

27 questions
Rochas Sedimentares
Presentation
•
11th Grade

27 questions
Biology Virus Lesson
Presentation
•
11th Grade

26 questions
Properties of Life LESSON
Presentation
•
10th - 12th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade
Discover more resources for Biology

16 questions
AP Biology: Unit 1 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
AP Biology: Unit 3 Review (CED)
Quiz
•
9th - 12th Grade

21 questions
AP Biology: Unit 6 Review (CED)
Quiz
•
9th - 12th Grade

14 questions
AP Biology: Unit 4 Review (CED)
Quiz
•
9th - 12th Grade

20 questions
Food Chains and Food Webs
Quiz
•
7th - 12th Grade

30 questions
AP Biology Unit 8 Review
Quiz
•
9th - 12th Grade

15 questions
Evidence of Evolution
Quiz
•
8th - 12th Grade

15 questions
Pedigrees
Quiz
•
7th - 12th Grade