

Boyle's Law
Presentation
•
Chemistry
•
11th Grade
•
Medium

Kelly Hutchens
Used 6+ times
FREE Resource
3 Slides • 10 Questions
1

Boyle's Law
2

Edpuzzle
You can open this webpage in a new tab.
3

Edpuzzle
You can open this webpage in a new tab.
4
Multiple Choice
5
Multiple Choice
1. A sample of oxygen gas occupies a volume of 250.L at 740. torr pressure. What volume will it occupy at 800. torr pressure?
266 L
245 L
231 L
217 L
6
Multiple Choice
7
Multiple Choice
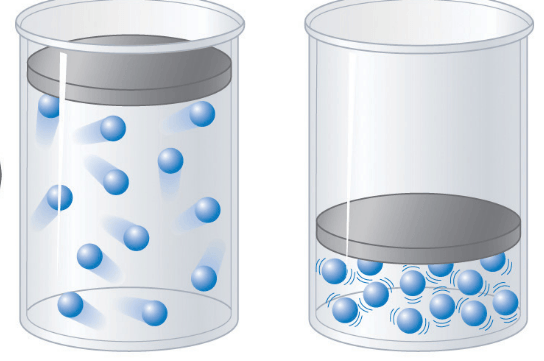
8
Multiple Choice
9
Multiple Choice
10
Multiple Choice
A gas with a volume of 4.0 L at 90.0 kPa expands until the pressure drops to 20.0 kPa. What is its new volume if the temperature doesn’t change?
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice

Boyle's Law
Show answer
Auto Play
Slide 1 / 13
SLIDE
Similar Resources on Wayground

10 questions
L2 Matter, Energy and Change
Presentation
•
10th - 12th Grade

10 questions
Conversions & Measurements notes
Presentation
•
10th - 11th Grade

8 questions
Kinetic Molecular Theory
Presentation
•
11th Grade

9 questions
Standing Waves
Presentation
•
11th Grade

10 questions
Planck's Equation
Presentation
•
11th Grade

10 questions
Fluida Statis As. Sumatif OTK
Presentation
•
11th Grade

12 questions
Cálculos estequiométricos
Presentation
•
11th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

34 questions
Nuclear Chemistry Review
Quiz
•
9th - 12th Grade

15 questions
Unit 9 solubility curves
Presentation
•
9th Grade - University

5 questions
DOL REC: Redox Reactions
Quiz
•
10th - 12th Grade

15 questions
IP REC: Redox Reactions
Quiz
•
10th - 12th Grade

39 questions
Nuclear Equations
Quiz
•
9th - 11th Grade