

CFE Review #1
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Justin Hall
Used 2+ times
FREE Resource
3 Slides • 30 Questions
1
CFE Review #1
Unit 1.2 and Unit 1.3
2
Unit 1.2 - Matter and Change
Phases of Matter (Chapter 2, Lesson 1)
Physical / Chemical Properties and Changes (Chapter 2, Lesson 2)
Elements, Compounds, and Mixtures (Chapter 2, Lesson 3)
Mixtures and Separations (Chapter 2, Lesson 4)
Standard: SC.912.P.8.2
3
Multiple Choice

Which state of matter has definite volume but not definite shape?
Solid
Liquid
Gas
4
Multiple Choice

Which state of matter has no definite size or shape?
Solid
Liquid
Gas
5
Multiple Choice
doesn't really change much
is easily reversible
6
Multiple Choice

Bubbling and steam escaping from boiling water is a
Physical change
Chemical change
7
Multiple Choice

Splitting logs into pieces with an axe to make firewood is a
Physical change
Chemical change
8
Multiple Choice

FIrewood burning in a fire is a
Physical change
Chemical change
9
Multiple Choice

Fizzy gas bubbles produced as a powder is added to a clear liquid is a
Physical change
Chemical change
10
Multiple Choice
11
Multiple Choice

12
Multiple Choice

13
Multiple Choice
14
Multiple Choice

A type of mixture that has the SAME composition in every part is
homogenous
heterogeneous
15
Unit 1.3 - Atomic Structure and the Periodic Table
Atomic Theory (Chapter 3, Lesson 1)
Atomic Structure (Chapter 3, Lesson 2)
Protons, Neutrons, and Electrons (Chapter 3, Lesson 3)
Development of the Periodic Table (Chapter 5, Lesson 1)
Groups, Periods, and Blocks of the Periodic Table (Chapter 5, Lesson 2)
Periodic Trends (Chapter 5, Lesson 3)
Standards: SC.912.P.8.3, SC.912.P.8.4, SC.912.P.8.5
16
Multiple Choice
17
Multiple Choice
Which element in Period 2 has the greatest atomic radius?
Be
C
Na
Li
18
Multiple Choice
19
Multiple Choice
Electronegativity __________ from left to right within a period and __________ from top to bottom within a group.
decreases, increases
increases, increases
increases, decreases
stays the same, increases
20
Multiple Choice
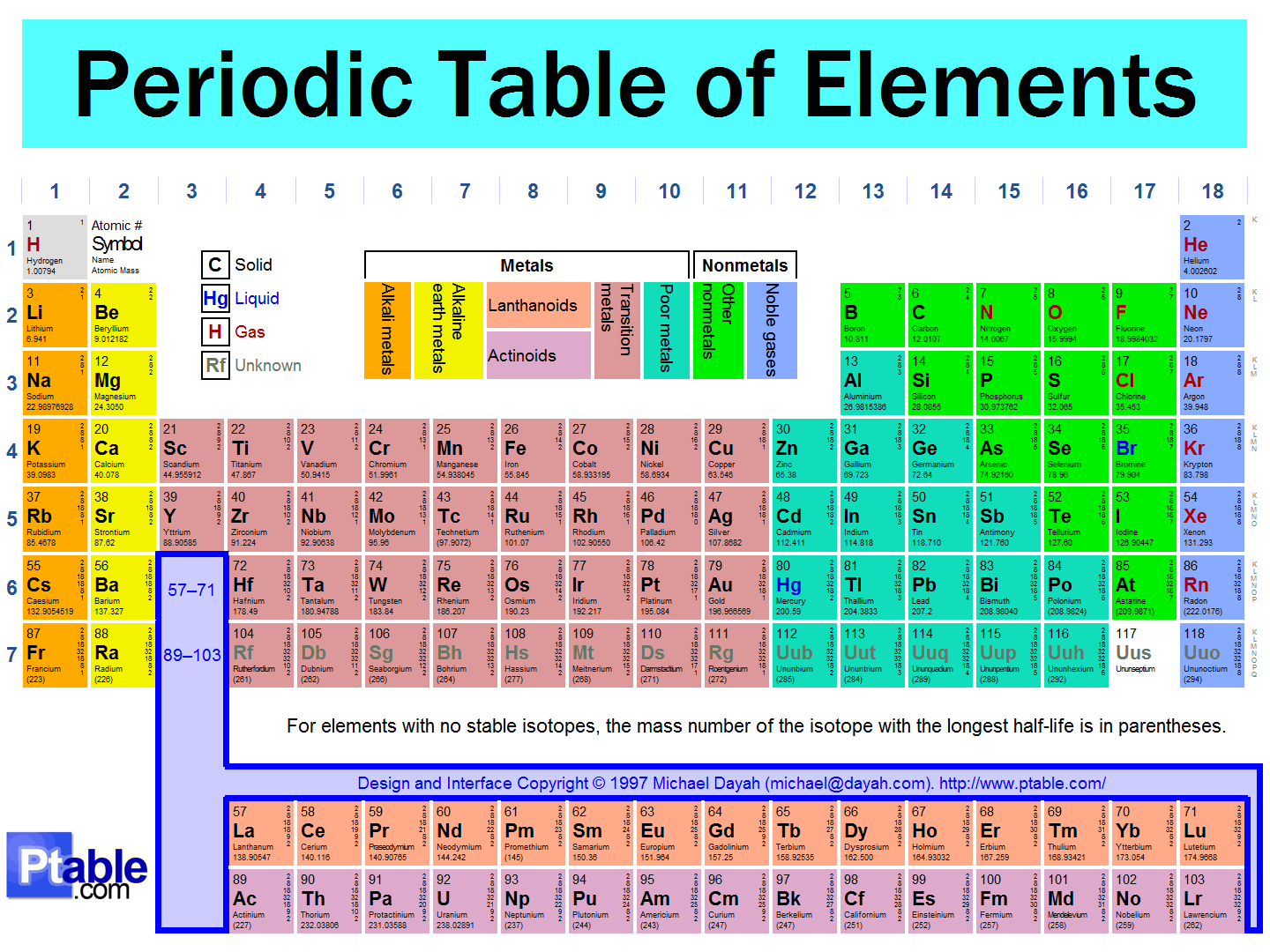
The vertical (up and down) columns in the Periodic Table are called
groups
towers
periods
atomic numbers
21
Multiple Choice
Which has the greater electronegativity:
H or F?
22
Multiple Choice
How many valence electrons does carbon have?
4
5
6
8
23
Multiple Choice
24
Multiple Choice
25
Multiple Choice

How many protons does sulfur have?
32
16
48
12
26
Multiple Choice
In a neutral atom, the number of protons is equal to the number of ____________.
energy levels
neutrons
neurons
electrons
27
Multiple Choice
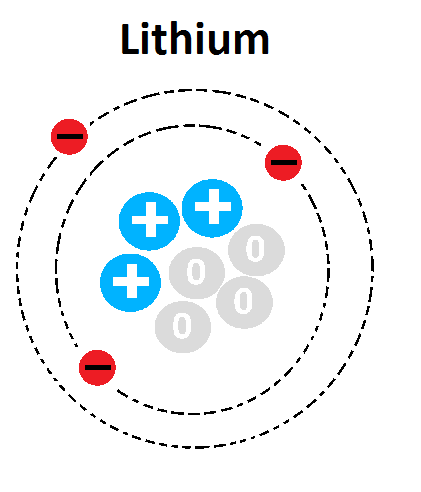
What is the atomic number of this atom?
1
3
4
7
28
Multiple Choice
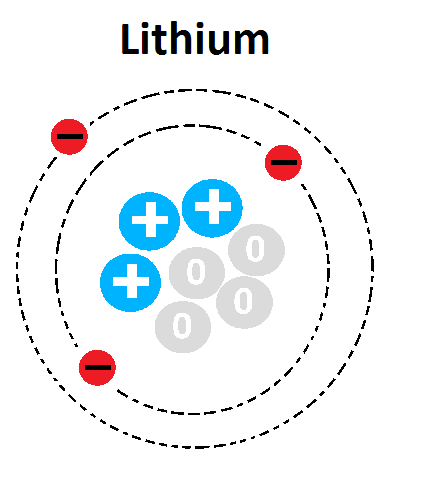
What is the mass number of this atom?
1
3
4
7
29
Multiple Choice
30
Multiple Choice
Add the number of electrons and protons together
Subtract the number of electrons from the number of protons
Subtract the number of protons from the mass number
Add the mass number to the number of electrons
31
Multiple Choice
Which of the following determines the identity of an element?
number of protons
atomic mass
number of neutrons
number of shells
32
Multiple Choice
Isotopes of an element have a different number of...
Protons
Neutrons
Electrons
Mass
33
Multiple Choice
CFE Review #1
Unit 1.2 and Unit 1.3
Show answer
Auto Play
Slide 1 / 33
SLIDE
Similar Resources on Wayground

27 questions
Moles - Part 2
Presentation
•
9th - 12th Grade

25 questions
Predicting Products
Presentation
•
9th - 12th Grade

26 questions
Chem LStd 1.1 Review - Mass, Volume
Presentation
•
9th - 12th Grade

26 questions
Notes on Compound Inequalities
Presentation
•
9th - 12th Grade

26 questions
Stoichiometry Lesson
Presentation
•
9th - 12th Grade

25 questions
Electron Configuration, Orbital Notation, & Dot diagrams
Presentation
•
9th - 12th Grade

27 questions
Bonding and Formulas
Presentation
•
9th - 12th Grade

22 questions
Accuracy v. Precision, Sig Figs, and Scientific Notation
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

30 questions
GVMS House Trivia 2026
Quiz
•
6th - 8th Grade
Discover more resources for Chemistry

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

20 questions
Interpreting Solubility Curves
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
10th Grade

43 questions
Acids and Bases Review
Quiz
•
10th - 11th Grade

20 questions
Thermochemistry Review
Quiz
•
10th Grade

25 questions
Unit 4 (Mid) Rumble
Quiz
•
10th Grade