

Solubility Curves
Presentation
•
Science, Chemistry
•
10th - 12th Grade
•
Practice Problem
•
Medium
Standards-aligned
Meghan RolesvilleHS
Used 406+ times
FREE Resource
12 Slides • 16 Questions
1
Solutions & Solubility
How to

2
What is a Solution?
Solution: a mixture that has the same composition, color, density & taste throughout
A solution has two parts:
> Solute = substance dissolved (less)
> Solvent = does the dissolving (more)
3
Multiple Choice
When making Kool-aid, the flavor powder would be the ____
solute
solvent
4
Multiple Choice
When making Kool-aid, the water would be the ____
solute
solvent
5
Concentration
Concentration: percent by volume of solute in solvent
Concentrated: large amount of solute in solvent
Dilute: small amount of solute in solvent
6
Multiple Choice

Which solution is the most concentrated?
A
B
C
D
E
7
Types of Solutions
Unsaturated - able to dissolve more solute at a given temperature (not full)
Saturated - contains all solute it can hold at a given temperature (full)
Supersaturated - contains more dissolved solute than necessary to make a saturated solution (unstable)
8
Multiple Choice

If this beaker can hold a maximum amount of 3.0 g of solute, is this beaker saturated or unsaturated?
unsaturated
saturated
9
Multiple Choice

If this beaker can hold a maximum amount of 3.0 g of solute, is this beaker saturated or unsaturated?
unsaturated
saturated
10
Multiple Choice
When a small amount of additional solute is added to an unsaturated solution, what happens to the number of dissolved particles? Do they go up, down, or stay the same?
up
down
stays the same
11
Multiple Choice
When a small amount of additional solute is added to a saturated solution, what happens to the number of dissolved particles? Do they go up, down, or stay the same?
up
down
stays the same
12
What is Solubility?
Solubility: the maximum amount of solute that will dissolve in a certain amount of solvent at a given temperature
> Solute = substance dissolved
> Solvent = does the dissolving
Example: ___grams of salt (NaCl) dissolved in 100g of water at ___℃
13
Temperature and Solubility
Temperature affects how much of the solute can be dissolved by the solvent.
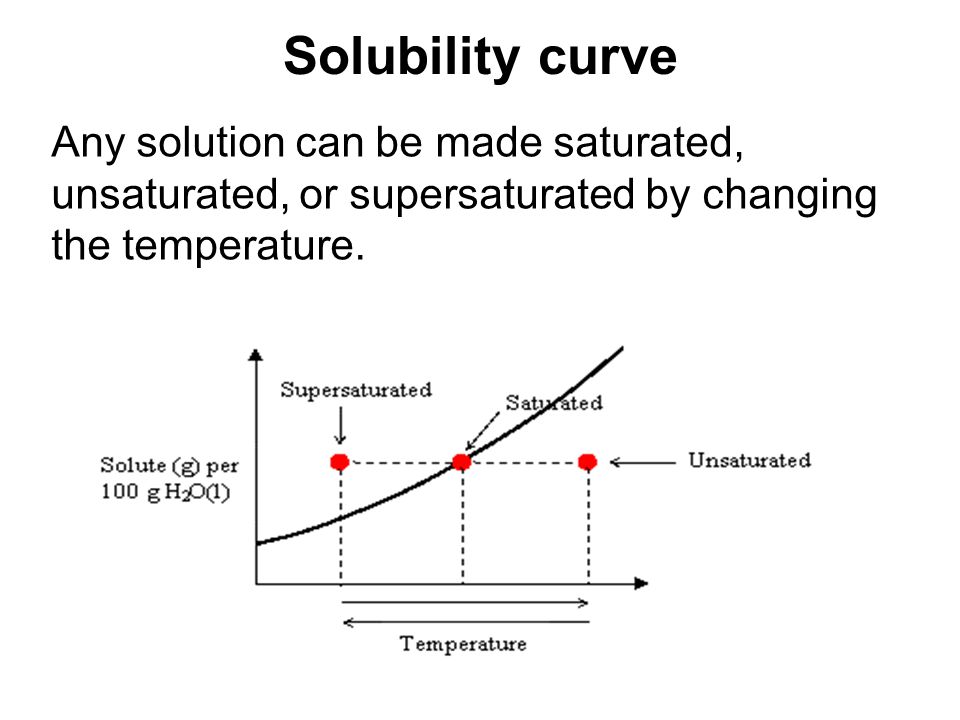
Solubility Curves: Are used to show how the solubility of a substance changes with temperature
14
15
Solubility Curve - Saturated
Any point on a line represents a saturated solution
In a saturated solution, the solvent contains the maximum amount of solute
Example: At 90℃, 40g of NaCl in 100g H2O represent a saturated solution.

16
Solubility Curve - Unsaturated
Any point below a line represents an unsaturated solution.
In an unsaturated solution, the solvent contains less than the maximum amount of solute.
Example: At 90℃, 30g of NaCl in 100g of H2O represents an unsaturated solution.
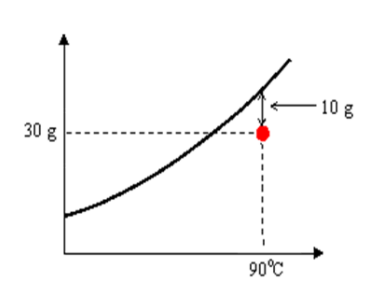
17
Solubility Curve - Supersaturated
Any point above a line represents a supersaturated solution.
In a supersaturated solution, the solvent contains more than the maximum amount of solute.
Example: At 90℃, 50g of NaCl in 100g H2O represents a supersaturated solution

18
To read the graph:
1 - Find the line for the substance
2 - The amount that dissolves at a given temperature is on the y-axis
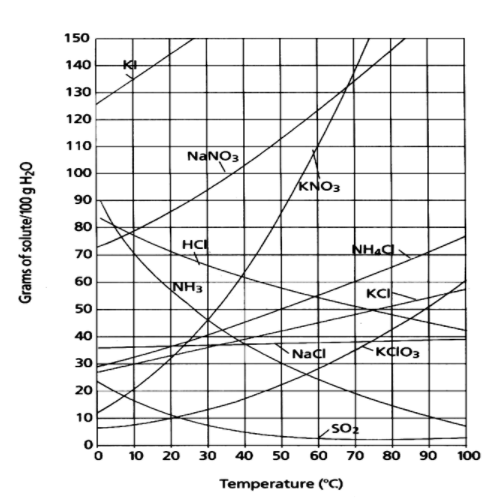
19
How much KNO3 dissolves in 100g H2O at 50℃
- Find the line for KNO3 (red)
- Find the temperature and follow up to the line for KNO3 (green)
- Read across to the y-axis and this is the answer (blue)
- About 87g of KNO3 will dissolve

20
Multiple Choice

How many grams of ammonium chloride (NH4Cl) will dissolve at 60°C?
70 g
55 g
40 g
21
Multiple Choice

How many grams of KNO3 can be dissolved at 30°C?
47 g
18 g
10 g
22
Multiple Choice

Is the following solution unsaturated, saturated, or supersaturated?
100g of NaNO3 at 30°C
unsaturated
saturated
supersaturated
23
Multiple Choice

How many grams of NaCl are required for the solution to become saturated at 50°C?
38 g
100 g
30 g
24
Multiple Choice

At what temperature would 60g of NH4Cl completely dissolve?
55°C
70°C
100°C
25
Multiple Choice

Is the following solution unsaturated, saturated, or supersaturated?
53g of NH3 at 20°C
unsaturated
saturated
supersaturated
26
Multiple Choice

Is the following solution unsaturated, saturated, or supersaturated?
10g of Ce2(SO4)3 at 10°C
unsaturated
saturated
supersaturated
27
Multiple Choice

Is the following solution unsaturated, saturated, or supersaturated?
125g of KNO3 at 60°C
unsaturated
saturated
supersaturated
28
Multiple Choice

Is the following solution unsaturated, saturated, or supersaturated?
65g of NH4Cl at 80°C.
unsaturated
saturated
supersaturated
Solutions & Solubility
How to

Show answer
Auto Play
Slide 1 / 28
SLIDE
Similar Resources on Wayground

22 questions
Metallic Bonds
Presentation
•
10th - 11th Grade

22 questions
Introduction to Molarity
Presentation
•
10th - 12th Grade

19 questions
Bonding Properties
Presentation
•
10th - 12th Grade

21 questions
12.3-4 Lymphatic System
Presentation
•
11th - 12th Grade

20 questions
⏳ENV_Recovery_Sustainable Behavior_4.28.23
Presentation
•
9th - 12th Grade

22 questions
4.5: Stoichiometry
Presentation
•
9th - 12th Grade

21 questions
BIOLOGI TINGKATAN 4 BAB 3
Presentation
•
11th Grade

23 questions
8.1 Introduction to Acids and Bases
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
Factors 4th grade
Quiz
•
4th Grade

10 questions
Cinco de Mayo Trivia Questions
Interactive video
•
3rd - 5th Grade

13 questions
Cinco de mayo
Interactive video
•
6th - 8th Grade

20 questions
Math Review
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Science

100 questions
Biology EOC Review
Quiz
•
9th - 12th Grade

38 questions
Biology EOC Review Game
Quiz
•
10th Grade

25 questions
Biology EOC review Quiz
Quiz
•
9th - 12th Grade

9 questions
EOC Domain 4: Evolution
Presentation
•
9th - 10th Grade

14 questions
EOC Domain 2: Genetics
Presentation
•
9th - 10th Grade

10 questions
EOC Domain 3: Taxonomy
Presentation
•
9th - 10th Grade

12 questions
EOC Domain 5: Ecology
Presentation
•
9th - 10th Grade

10 questions
Exploring Fusion and Fission in Nuclear Chemistry
Interactive video
•
6th - 10th Grade