

3.3 Average Atomic Mass
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Standards-aligned
Katherine Fehrenbach
Used 3+ times
FREE Resource
8 Slides • 3 Questions
1

2

3

4

5

6

7

8

9
Multiple Choice
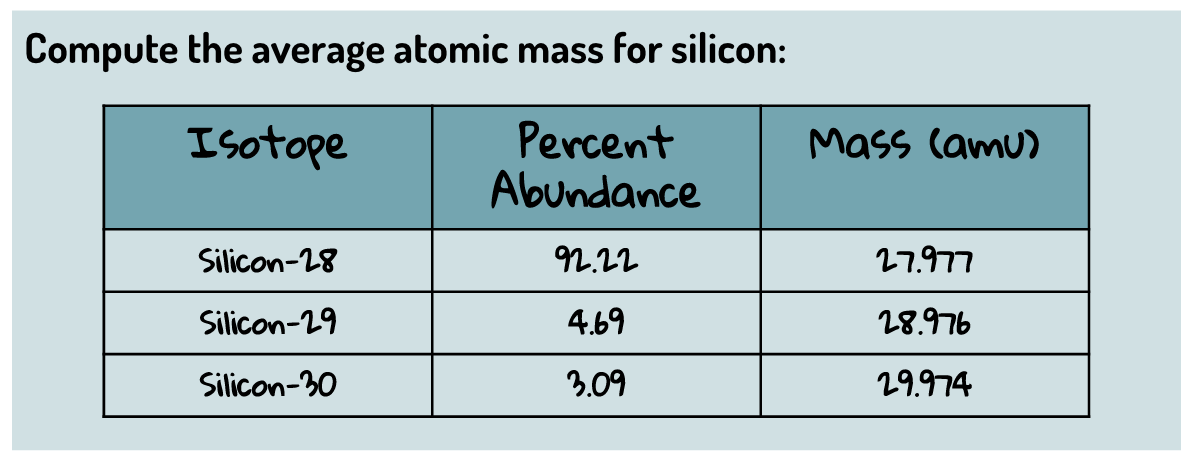
What is the average mass of this element?
10
Multiple Choice
11
Multiple Choice

Show answer
Auto Play
Slide 1 / 11
SLIDE
Similar Resources on Wayground

11 questions
Significant Figures
Presentation
•
10th - 12th Grade

9 questions
Y = MX + B Review
Presentation
•
9th - 12th Grade

12 questions
Molar Conversions
Presentation
•
9th - 12th Grade

6 questions
MAC 2 | Lesson 5-7 | Wrap it Up
Presentation
•
KG

6 questions
Electron configuration
Presentation
•
9th - 12th Grade

10 questions
Domain 2 Lesson 3
Presentation
•
9th - 12th Grade

10 questions
Writing an equation from a graph
Presentation
•
9th - 12th Grade

8 questions
Thermochemistry Notes Part 2
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

20 questions
Figurative Language Review
Quiz
•
6th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

10 questions
Identify Fractions, Mixed Numbers & Improper Fractions
Quiz
•
3rd - 4th Grade
Discover more resources for Chemistry

25 questions
Waygrounds 5/20 PS.8.1.2
Quiz
•
9th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade

15 questions
Types of chemical reactions
Quiz
•
9th - 12th Grade

32 questions
Acids/Bases, Electrochem, Orgo
Quiz
•
9th - 12th Grade