

7.3.3-The Behavior of Gases
Presentation
•
Mathematics
•
8th Grade
•
Medium
+8
Standards-aligned
Jessica Freeman
Used 6+ times
FREE Resource
34 Slides • 51 Questions
1

2

3

4

5

6

7
Kinetic Molecular Theory

Review
8

The states of matter : All matter is composed of tiny particles that are always in motion.
The Kinetic Molecular Theory
9
Multiple Choice
The kinetic theory states
Particles won't move if you don't apply energy to it.
Particles only move in liquids and gases.
Particles are always in motion.
At the same temperature the object that has the most mass heats quicker.
10

Definition Shape
Definite Volume
Little movement
Example:
ICE
Solids
11
Multiple Choice
12

NO Definition Shape
Takes the shape of the container.
Definite Volume
Molecules have a "FLOW"
Medium Kinetic Energy
Example:
Water
Liquids
13
Multiple Choice
14
Multiple Choice
15

NO definition Shape
Takes the shape of the container.
NO definite Volume
Fills the volume of the container
Molecules move FAST and Random
High Kinetic Energy
Example:
Steam
Gas
16
Change the TEMPERATURE!
How do we move from one state to the next?

High Kinetic Energy
Low Kinetic Energy
17
Multiple Choice
18
Multiple Choice
19
Multiple Choice
20
Multiple Choice
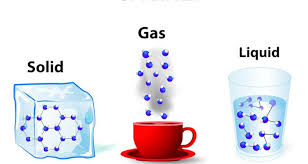
21
Multiple Choice
As the _________________ of a gas increases, the kinetic energy of the particles increases. The particles move farther apart, and volume increases.
speed
height
temperature
area
22
Multiple Choice
Gases assume the volume and shape of their containers
TRUE
FALSE
23
Multiple Choice

State of matter that can fill containers with no fixed shape or volume.
Solid state
Liquid state
Gas state
Plasma state
24

25

26
Pressure
Pressure: The amount of force applied per unit of area.
In gases, as pressure increases, volume decreases.
Why?
When the particles in a gas are compressed, they have less space to move around. As a result, the particles start to collide with one another more often — this increases pressure!

27

28
Boyle's Law
Robert Boyle (1627-1691) was a British scientist. He was the first to describe the relationship between volume and gases. He also proved that gases are made up of atoms.
Boyle's Law
P1V1 = P2V2
Pressure and Volume have an inverse (opposite) relationship when temperature is held constant.
The pressure of a gas increases when the volume decreases.
The pressure of a gas decreases when the volume increases.



29
Graphing Boyle's Law
Pressure is graphed on the x-axis
Volume is graphed on the y-axis
As pressure increases, volume decreases. This is true only if the temperature of the gas is held constant.

30
Fill in the Blanks
Type answer...
31
Fill in the Blanks
Type answer...
32

33

34
Boyle's Law in Action
Air pressure and flying
On the ground, your middle ear and the air outside have equal pressure.
As the airplane takes off and increases in altitude, the air pressure outside decreases relative to your middle ear.
In response to the decrease in pressure, the trapped air in your middle ear increases in volume, which causes pain.

35
Boyle's Law Examples
Bike pump: When you pump air into a tire, the gas molecules inside the tire get compressed and packed closer together. This increases the pressure of the gas, and it starts to push against the walls of the tire. You can feel how the tire becomes pressurized and tighter.
Soda Bottles: To get carbon dioxide gas into the liquid, the whole bottle is usually pressurized with gas. As long as the bottle is closed, it is very hard to squeeze, as the gas is confined to a small space and pushes against the bottle's walls. When you remove the cap, however, the available volume increases and some of the gas escapes. At the same time its pressure decreases.
36
Boyle's Law Formula
P1 X V1= P2 X V2
P=pressure
V=volume
*Temperature must be constant
A 200mL can of spray with a pressure of 20kPA is run over by a car and flattened to a volume of 10 mL. What is the new pressure? Should it be greater or less than the initial 20kPA (kilo Pascal)?
20 X 200 = ___ X 10
37
Multiple Choice
What is the relationship between volume and pressure?
directly related: if one goes up, the other goes up too
inversely related: if one goes up, the other goes down
no relationship
38
Multiple Choice
If volume increases, the pressure __________
increases
decreases
stays the same
39

40
Multiple Choice

41
Multiple Choice

42
Multiple Choice
A flask contains 155 cm3 of hydrogen at a pressure of 22.5 kPa. Under what pressure would the gas have a volume of 90.0 cm3 at the same temperature?
38.8 kPa
43.9 kPa
43

44
Charles's Law: Volume and Temperature are DIRECTLY RELATED
Charles’s law or the law of volumes is an ideal gas law that states that the volume and temperature of a fixed amount of gas are proportional at constant pressure. Doubling the temperature of a gas doubles its volume. Halving the temperature of a gas halves its volume. The law takes its name from French scientist Jacques Charles, who formulated the law in the 1780s.

45
Charles's Law Examples
The official rules of the NFL require footballs to be inflated to a gauge pressure between 12.5 to 13.5 pounds per square inch. The rules do not specify the temperature at which such measurement is to be made. Thus, if a football were inflated to the minimum pressure of 12.5 psi at room temperature, the pressure would drop below the minimum as the gases inside cooled to a colder temperature on the playing field.

AFC Playoff Game Results
New England Patriots: 45
Indianapolis Colts: 7
Outside Temperature: 52*F
46
Charles's Law Examples
Car tires have lower PSI during the winter
Deodorant spray bottle warning labels: "pressurized container, protect it from sunlight. Do not expose to temperature exceeding 50°C”
Hot Air Balloon-when the gas is heated, it expands, and becomes less dense and the balloon is lifted in the air. The warm is less dense than the cold air, which means that it is lighter than the cold air. Also, the warm air has less mass per unit volume
47
Multiple Choice
If temperature decreases, volume ________
Think: What could happen to a balloon when it's taken outside on a cold day?
increases/gets bigger
decreases/gets smaller
stays the same
48
Multiple Choice
What is the relationship between temperature and volume?
related: if one goes up, the other goes up
inversely related: if one goes up, the other goes down
49

50

51

52
Multiple Choice

53
Multiple Choice

Which law?
Boyle
Charles
Landis
Boykin
54
Multiple Choice
55
Charles's Law
relates temperature to volume.
States "For a fixed amount of gas at a constant pressure, the volume of the gas increases as the gas's temperature increases.
Because of Charles's law, an inflated balloon will pop when it gets too hot.
56

57
Multiple Choice
58

59
Multiple Choice
Temperature and pressure are ____________ proportional.
directly
inversly
synergistically
60
Multiple Choice
P1/T1 = P2/T2
P1V1 = P2V2
V1/T1 = V2/T2
PV = nRT
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
pressure & amount of gas
volume & amount of gas
64
Multiple Choice
65
Multiple Choice
66
Multiple Choice
67
Multiple Choice
Boyles Law Is A
Directly Proportional Relationship
Inversely Proportional Relationships
68
Multiple Choice
Boyles Law Tells That
The More The Volume The Less The Temperature
The More The Temperature The Less The Pressure
The More The Volume The More The Pressure
The More The Pressure The Less The Volume
69
Multiple Choice
What type of relationship to pressure and volume have?
direct
no relationship
inverse
I don't know
70
Multiple Choice
When Volume increases then Pressure must...
Increase
Decrease
71
Multiple Choice
When Pressure increases then the Volume must...
Increase
decrease
72
Multiple Choice
According to Charles Law, if Temperature of a gas increases Volume will also....
Increases
Decreases
Stays the same
73
Multiple Choice
According to Charles Law, the relationship between Temperature and Volume can best be described as....
Inverse Relationship
No Relationship
Direct Relationship
74
Multiple Choice
Which of these is NOT a unit of pressure?
atm
torr
psi
ml
75
Multiple Choice
Which of these is NOT in Boyle's law (because it stays the same?)
Pressure
Volume
Temp
76
Multiple Choice
Which of these is NOT in CHARLES's law (because it stays the same?)
Pressure
Volume
Temp
77
Multiple Select
One goes up, one goes down is which of the following: PICK TWO
Inverse proportion
Charles' Law
Direct Proportion
Boyles' Law
78
Multiple Select
BOTH goes up, BOTH goes down is which of the following: PICK TWO
Inverse proportion
Charles' Law
Direct Proportion
Boyles' Law
79
Multiple Choice
Which gas law would you use to solve the following:
At 27.00 °C a gas has a volume of 6.00 L. What will the volume be at 150.0 °C?
Boyles
Charles
Lussacs
Combined
80
Multiple Choice
81
Multiple Choice

82
Multiple Choice

Which law?
Charles
Boyle
Diegel
Mitchell
83
Multiple Choice
84
Multiple Choice

85
Multiple Choice


Show answer
Auto Play
Slide 1 / 85
SLIDE
Similar Resources on Wayground

80 questions
Unit 1- Economic Systems
Presentation
•
9th Grade

80 questions
Evolution Intro and Natural Selection
Presentation
•
9th Grade

80 questions
Solve Quadratic Inequalities
Presentation
•
9th Grade

79 questions
8.3.3-Describing Circuits
Presentation
•
9th Grade

79 questions
7.SP.A.1-Sampling the Truth: Making Smart Inferences from Data
Presentation
•
7th Grade

77 questions
Ch.1 LA Culture Sect. 2 Festivals
Presentation
•
8th Grade

81 questions
Review2025
Presentation
•
8th Grade

79 questions
D157 Activity: iLearn Practice
Presentation
•
7th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Mathematics

48 questions
8TH GRADE MATH FAST REVIEW
Quiz
•
8th Grade

20 questions
summer trivia
Quiz
•
8th Grade

20 questions
Multi Step Equations with Variables on Both Sides
Quiz
•
8th Grade

20 questions
Scatter Plots and Line of Best Fit
Quiz
•
8th Grade

20 questions
Pythagorean Theorem Review
Quiz
•
8th Grade

12 questions
8th U8L8 Finding Unknown Side Lengths
Quiz
•
8th Grade

55 questions
8th Grade Math Review
Quiz
•
8th Grade

20 questions
Slope from a Graph
Quiz
•
8th Grade