

HC CH 9.3 Qzz
Presentation
•
Chemistry
•
9th Grade
•
Medium
Robert Oliver
Used 1+ times
FREE Resource
21 Slides • 16 Questions
1

2

3

4
Multiple Choice

What is true of a stoichiometric mixture?
all reactants are turned into products
at least one reactant has "leftover" particles
all reactants have "leftover particles
all products are turned into reactants
5

6
Multiple Choice

What is true of a limiting reactant mixture?
all reactants turn into products
at least one reactant has unused particles after the reaction
all reactants have unused particles after the reaction
all products are turned into reactants
7

8
Multiple Choice

What do we call the reactant that runs out first in a reaction?
limiting reactant
limiting product
noble reactant
unfulfilled reactant
noble product
9
Multiple Choice

What is the limiting reactant in this reaction?
H2
N2
NH3
H2O
10

11
Multiple Choice

What is the limiting reactant in this reaction?
12

13
Multiple Choice
Which of the following reaction mixtures could produce the greatest amount of product? Each involves the reaction symbolized by the equation:
2H₂ + O₂ → 2H₂O
2 moles of H₂ and 2 moles of O₂
2 moles of H₂ and 3 moles of O₂
2 moles of H₂ and 1 mole of O₂
3 moles of H₂ and 1 mole of O₂
Each produce the same amount of product.
14

15
Multiple Select

What information do you need to know in order to determine the mass of product that will be produced?
Balanced equation
Molar masses of A, B and the product
rate of the reaction
states of matter of reactants and product
16

17

18
Labelling
The molar mass of A is 10.0 g/mol.
If you have 10.0 g of A, how many moles do you have?
1 mol A
1
100
10.0 g A
19

20

21
Multiple Choice

What is the limiting reactant in this reaction?
A
B
C
There is none.
(stoichiometric mixture)
22

23
Labelling
A + 3B → 2C
Given this equation, how many moles of C will be produced from 1.00 moles of A and 0.500 moles of B?
0.500
3 mol B
0.333
2.00
1 mol A
2 mol C
24

25

26

27
Multiple Choice
What is the theoretical yield?
The actual amount of product formed in a reaction.
The maximum amount of product formed when the limiting reactant is completely consumed.
The percentage of the actual yield to the theoretical yield.
The amount of reactants used in a reaction.
28
Multiple Choice
How is percent yield calculated?
Actual yield / Theoretical yield x 100%
Theoretical yield / Actual yield x 100%
Actual yield + Theoretical yield x 100%
Theoretical yield - Actual yield x 100%
29
Multiple Choice
What is usually true about the actual yield of a reaction?
It is always equal to the theoretical yield.
It is usually greater than the theoretical yield.
It is usually less than the theoretical yield.
It is not related to the theoretical yield.
30

31

???
32
Fill in the Blanks

33

34

= ???
35
Categorize


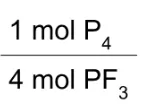
Which resource was used to determine the conversion factors?
36
Fill in the Blanks

37


Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

33 questions
Electromagnetic Waves
Presentation
•
9th Grade

31 questions
3D Half Life
Presentation
•
9th Grade

28 questions
Periodic Table Review
Presentation
•
8th Grade

30 questions
Solutions, suspensions and colloids
Presentation
•
9th Grade

31 questions
Lesson: Angled Projectile Motion
Presentation
•
9th Grade

29 questions
Intro to Linear Equations
Presentation
•
9th Grade

29 questions
Year 9 Science 9.4 The Reactivity Series
Presentation
•
9th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade