

B.4.2 Lesson 4 Thermodynamic Processes
Presentation
•
Physics
•
11th Grade
•
Easy
Lester Orellana
Used 1+ times
FREE Resource
4 Slides • 33 Questions
1
Multiple Choice
2

3
Multiple Choice
4
Multiple Choice

Process B on the PV diagram is an ______________ process.
adiabatic
isobaric
isochoric
isothermal
5
Multiple Choice
A thermodynamic system where no exchange of heat takes place between system and surrounding is...
Isothermal process
Adiabatic process
Isobaric process
Isochoric process
6
Multiple Choice
7
Multiple Choice
8
Multiple Choice
A thermodynamic system where no exchange of heat takes place between system and surrounding is...
Isothermal process
Adiabatic process
Isobaric process
Isochoric process
9
Multiple Choice
Heating a closed, steel container is an example of an
Adiabatic process
Isovolumetric process
Isothermal process
Isobaric process
10
Multiple Choice
For an isovolumetric process, which of the following statements is correct?
a. Work, heat, and internal energy all undergo changes.
b. Work and heat balance each other, so that there is no change in internal energy.
c. No energy is transferred as heat; internal energy change is due to work.
d. No work is done; internal energy change is due to heat.
11
Multiple Choice
Symbol that indicates that energy is released.
U
Q
-Q
W
12
Multiple Choice
In an adiabatic process, what equals zero?
Q
W
ΔU
T
13
Multiple Choice
Pistons moving in a car is an example of an adiabatic process
True
False
14
Multiple Choice
In Isothermal process which property remains constant
Volume
Pressure
Concentration
Temperature
15
Multiple Choice
What is the name of the process in which pressure remains constant.
Isochoric process
Adiabatic process
Isobaric process
Isolated process
16
Multiple Choice
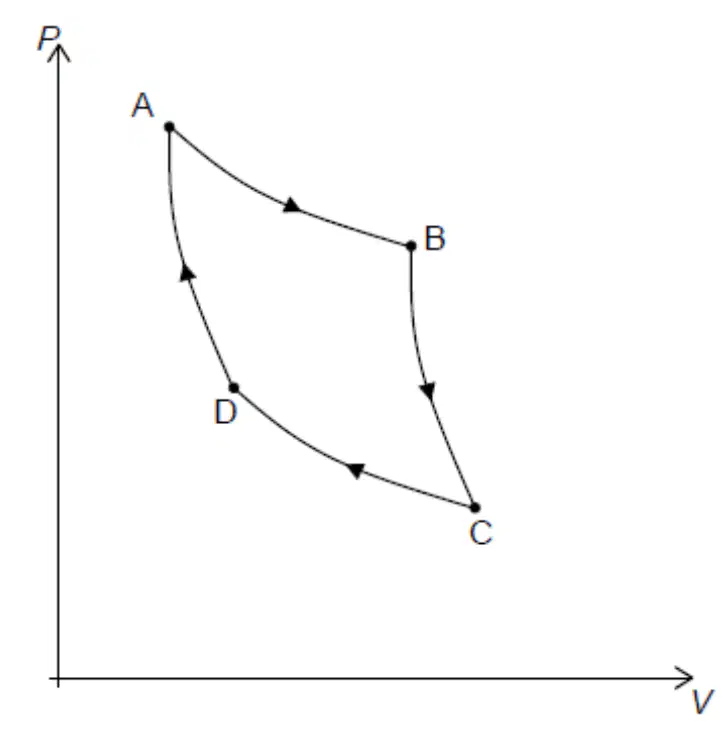
The picture represents a Carnot Cycle.
what type of process is A-B
Isothermal Expansion
Isothermal Compression
Adiabatic Expansion
Adiabatic Compression
17
Multiple Choice

The picture represents a Carnot Cycle.
what type of process is B-C
Isothermal Expansion
Isothermal Compression
Adiabatic Expansion
Adiabatic Compression
18
Multiple Choice

The picture represents a Carnot Cycle.
what type of process is C-D
Isothermal Expansion
Isothermal Compression
Adiabatic Expansion
Adiabatic Compression
19
Multiple Choice

The picture represents a Carnot Cycle.
what type of process is D-A
Isothermal Expansion
Isothermal Compression
Adiabatic Expansion
Adiabatic Compression
20
Multiple Choice

The picture represents a Carnot Cycle.
Which process keeps the same temperature but compresses the volume.
A-B
B-C
C-D
D-A
21
Multiple Choice

The picture represents a Carnot Cycle.
Which process represents partially the 1st Law of
Thermodynamic –Q=–∆V.
A-B
B-C
C-D
D-A
22
Multiple Choice
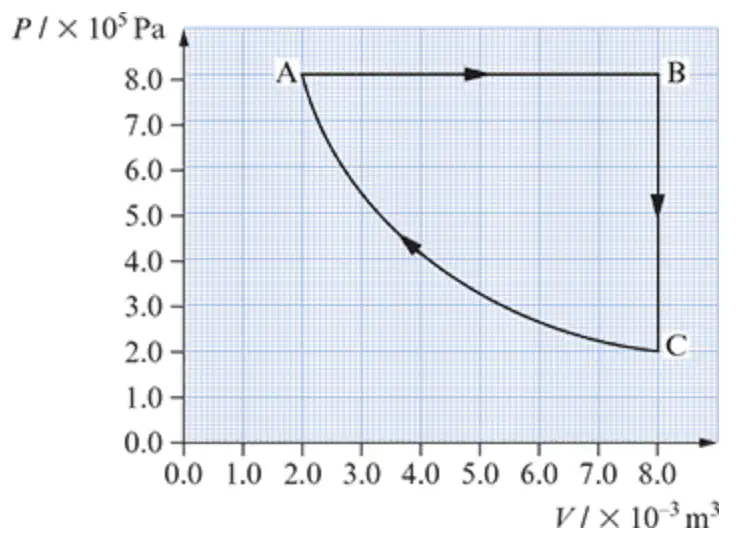
Which process is isobaric?
A-B
B-C
C-A
None
23
Multiple Choice

Which process shows +W?
A-B
B-C
C-A
None
24
Multiple Choice

Which process shows +∆V?
A-B
B-C
C-A
None
25
Multiple Choice

Which process shows ∆U=0?
A-B
B-C
C-A
None
26
Multiple Choice

Which process shows ∆Ek gas=0?
A-B
B-C
C-A
None
27
Multiple Choice

Which process shows –∆V?
A-B
B-C
C-A
None
28
Multiple Choice

Which process shows TB=TC?
A-B
B-C
C-A
None
29
Multiple Choice
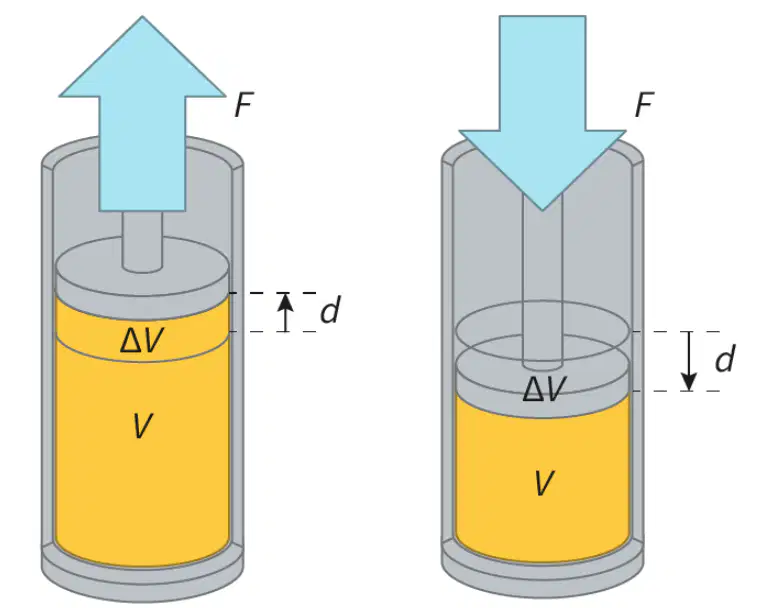
Isolated system.
The picture on the left represents:
Negative work by the gas inside the cylinder
Positive work by the gas inside of cylinder
Positive work by the surroundings.
30
Multiple Choice

Isolated System.
The picture on the left represents:
+∆V
+W
+∆P
–∆V
–∆P
31
Multiple Choice

Isolated System.
The picture on the right represents that the work is done on the gas:
+W
+∆P
–W
–∆P
32
Multiple Choice
What best describes an adiabatic process?
Heat exchange occurs with the surroundings
Heat exchange occurs within the system
No heat exchange occurs with the surroundings
No work is performed by the system
33
Explanation Slide...
An adiabatic process is one where there is no heat exchange between the system and its surroundings. This means that no heat enters or leaves the system during the process.
34
Multiple Choice
In an adiabatic expansion of an ideal gas, what happens to the temperature?
Increases
Decreases
Remains constant
None
35
Explanation Slide...
During an adiabatic expansion, the gas expands without any heat being added or removed. As the gas does work on the surroundings, it loses internal energy, leading to a decrease in temperature.
36
Multiple Choice
Which of the following processes is most likely to approximate an adiabatic process?
A well-insulated gas cylinder expanding quickly
A gas cylinder with controlled heat exchange
A gas cylinder expanding slowly with constant heat addition
A gas cylinder in contact with an ice bath
37
Explanation Slide...
A well-insulated gas cylinder expanding quickly is more likely to approximate an adiabatic process because the insulation prevents heat exchange, and the rapid expansion reduces the time available for heat transfer.
Show answer
Auto Play
Slide 1 / 37
MULTIPLE CHOICE
Similar Resources on Wayground

26 questions
Metric System Conversions
Presentation
•
11th - 12th Grade

31 questions
Polarity and Intermolecular Forces
Presentation
•
11th - 12th Grade

34 questions
Le subjonctif
Presentation
•
11th Grade

28 questions
Unit 8 coulomb's law day 2
Presentation
•
11th Grade

32 questions
Trắc nghiệm ôn tập HK2 Vật lí 10
Presentation
•
10th Grade

35 questions
Events of World War II
Presentation
•
11th Grade

33 questions
Run-on Sentences
Presentation
•
10th Grade

33 questions
11QT Static electricity and electric field
Presentation
•
11th Grade
Popular Resources on Wayground

15 questions
Grade 3 Simulation Assessment 1
Quiz
•
3rd Grade

22 questions
HCS Grade 4 Simulation Assessment_1 2526sy
Quiz
•
4th Grade

16 questions
Grade 3 Simulation Assessment 2
Quiz
•
3rd Grade

19 questions
HCS Grade 5 Simulation Assessment_1 2526sy
Quiz
•
5th Grade

17 questions
HCS Grade 4 Simulation Assessment_2 2526sy
Quiz
•
4th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

24 questions
HCS Grade 5 Simulation Assessment_2 2526sy
Quiz
•
5th Grade

20 questions
Math Review
Quiz
•
3rd Grade