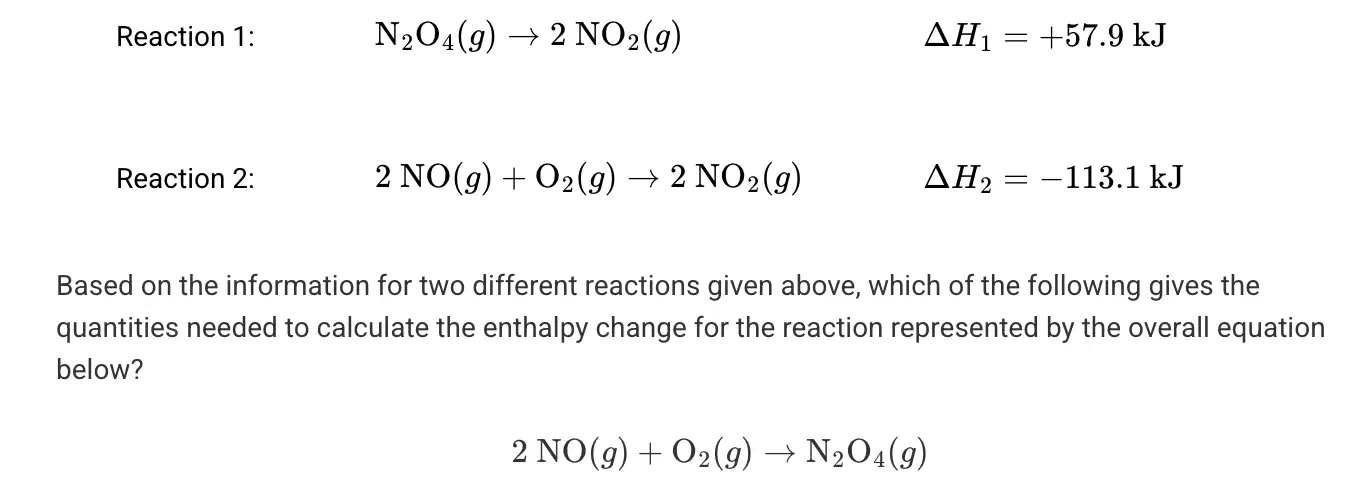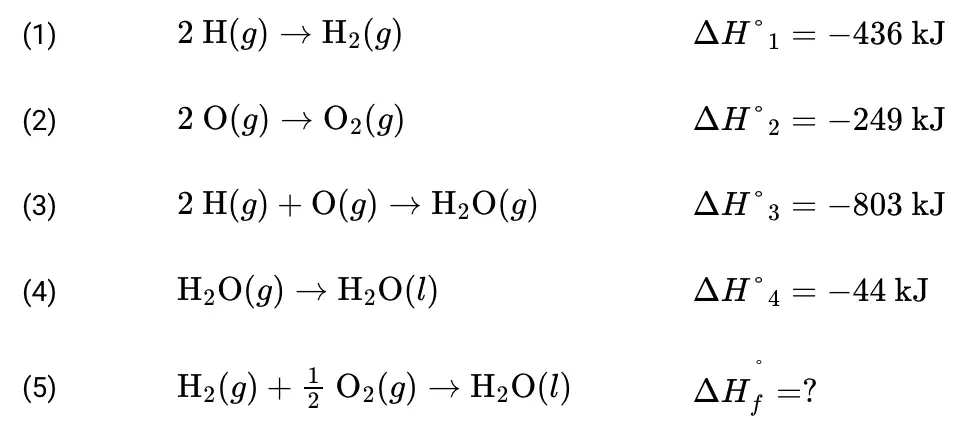6.8-6.9 Enthalpy of Formation and Hess's Law
Presentation
•
Chemistry
•
9th - 12th Grade
•
Practice Problem
•
Hard
•
NGSS
HS-PS1-4, HS-PS3-1, HS-PS1-2
Standards-aligned
Katherine Fehrenbach
FREE Resource
18 Slides • 8 Questions
1
2
Fill in the Blanks
3
Fill in the Blanks
4
5
6
7
8
9
10
11
12
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice
16
17
18
19
20
21
22
23
24
Multiple Choice
25
Multiple Choice
26
Multiple Choice
Show answer
Auto Play
Slide 1 / 26
SLIDE