

2-20-25 types of bonding slides 1-22
Presentation
•
Science
•
8th Grade
•
Medium
Standards-aligned
. Ade
Used 15+ times
FREE Resource
23 Slides • 45 Questions
1

2

3

4

5
Multiple Choice
fifty eleven
6
Multiple Choice
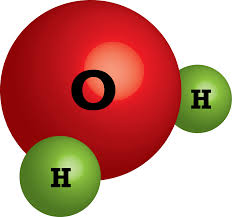
Determine the number of Hydrogen (H) atoms are in the following chemical formula: 3H2O
4
3
2
6
7
Multiple Choice
How many atoms are there TOTAL in:
H2SO4
6
5
7
3
8

9
Multiple Select

the EQ essential question for this lesson is what do ___, _____ & ______ have to do with atom bonding. 3 answers.
craig
smokey
re re
pac
D Bo, "that's my bike punk"
10

11
Multiple Choice
What is a chemical bond?
A weak force between molecules
A strong attractive force between atoms or ions in a molecule or compound
A type of chemical reaction
A process of breaking down compounds
12

13
Multiple Choice
What is the main reason behind bond formation?
Color
Stability
Size
Shape
habitability or outer layer
14
Multiple Select
How can a chemical bond be formed?
By heating the atoms
By sharing electrons
By mixing different compounds
By cooling the molecules
By transferring e-
15
Fill in the Blanks

16

17

18

19
Multiple Choice
Which element is an exception to the Octet Rule, having only 2 electrons in its outer shell? baby belly
Helium
Neon
Argon
Krypton
Li
20
Multiple Choice

What type of bond is formed when electrons are transferred between atoms?
Covalent bond
Ionic bond
Metallic bond
Hydrogen bond
Co Bailent
21
Multiple Choice

type of bond involves sharing of electrons between atoms?
Ionic bond
Covalent bond
Metallic bond
Hydrogen bond
CoVeneint bond
22
Multiple Choice

What are the two main types of chemical bonds mentioned?
north polar and south polar bonds
Ionic Bond and Covalent Bond
Van der Waals Bond and Peptide Bond
Polar Bond and Nonpolar Bond
ionate and covalent
23
Multiple Choice
What is an ionic bond?
Attraction between two oppositely charged ions
Sharing of electrons between atoms
Attraction between two like-charged ions
Sharing of protons between atoms
Attraction between two oppositely charged atoms
24
Multiple Select

What is a cation?
more than 1 correct choice
A negatively charged ion
A positively charged ion
elements that is be in Group 1 or 2
An ion formed by nonmetals
Na with 11p & 10 e-
25
Multiple Choice
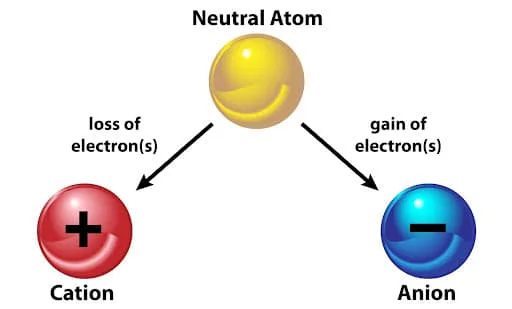
How is an anion formed?
By losing an electron
By gaining a proton
By gaining an electron
By losing a proton
by gaining e power to negate the proton orbital string theory
26
Multiple Select

What type of elements typically form cations?
Nonmetals
Metalloids
Metals
halogens
any alkaline group or family 2 elements
27

28

29
30
Multiple Choice

molecule formula
31
Multiple Choice

32
Multiple Choice
Is H an element or a molecule?
element
molecule
33
Multiple Choice

34
Multiple Choice
35
Multiple Select

choose all that apply
element
compound
molecule
water
hydrogen peroxide
36
Multiple Select

choose all that apply
only 1 element present
compound
molecule
water
Jayla made sum chicken but forgot the hotsauce.
37

38

39
Multiple Choice

What is a characteristic of a nonpolar covalent bond?
Electrons are shared unequally
Electrons are shared equally
Usually different atoms
Involves ionic bonding
40
Multiple Choice

What is a characteristic of a polar covalent bond?
Electrons are shared equally between atoms.
Electrons are transferred completely from one atom to another.
Electrons are shared unequally between two different atoms.
Electrons are not involved in bonding.
41
Multiple Choice
What type of bond is formed when chlorine shares a pair of electrons?
Ionic bond
Metallic bond
Hydrogen bond
Covalent bond
42
Multiple Choice
How many valence electrons does oxygen have?
2
4
6
8
43
Multiple Choice
How many electrons does oxygen share to form a covalent bond?
1
2
3
4
44

45

46

47

48
Multiple Choice

What type of bond is "RED" demonstrating with the desire to share the item?
Covalent bond
Ionic bond
Metallic bond
Hydrogen bond
49
Multiple Choice
Big guy D-BO best represents what type of bond?
Ionic bond
Covalent bond
Metallic bond
Hydrogen bond
50

51
Multiple Choice
Can covalent compounds conduct electricity?
Yes
No
Only in solid form
Only in liquid form
52
Multiple Choice
Are ionic compounds soluble in water?
No
Yes
Only in hot water
Only in cold water
53

54

55

56
Multiple Choice
What type of elements form ionic compounds?
Nonmetal only
Metal and nonmetal
Metal only
Nonmetal and metalloid
57
Multiple Choice
58
Multiple Choice
59
Multiple Choice
K1+ S2-
60
Multiple Choice
Al3+ O2-
61
Multiple Choice
62
Multiple Choice
63
Multiple Choice
64
Multiple Choice
65
Multiple Choice
Na1+ F1-
66
Multiple Choice
67
Multiple Choice
68
Multiple Choice

What is the melting point characteristic of covalent compounds?
High
Low
Medium
Variable
that mid grade

Show answer
Auto Play
Slide 1 / 68
SLIDE
Similar Resources on Wayground

61 questions
1.1 Metals and non metals
Presentation
•
7th Grade

65 questions
8.1 Exothermic Reaction
Presentation
•
7th Grade

68 questions
Chemistry Review
Presentation
•
8th Grade

63 questions
Middle School Chemistry Review
Presentation
•
8th Grade

63 questions
Chemistry Review
Presentation
•
8th Grade

62 questions
Classification of Matter
Presentation
•
9th Grade

67 questions
Physical and Chemical Change
Presentation
•
8th Grade

60 questions
Midterm Review
Presentation
•
7th - 8th Grade
Popular Resources on Wayground

20 questions
"What is the question asking??" Grades 3-5
Quiz
•
1st - 5th Grade

20 questions
“What is the question asking??” Grades 6-8
Quiz
•
6th - 8th Grade

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

34 questions
STAAR Review 6th - 8th grade Reading Part 1
Quiz
•
6th - 8th Grade

20 questions
“What is the question asking??” English I-II
Quiz
•
9th - 12th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

47 questions
8th Grade Reading STAAR Ultimate Review!
Quiz
•
8th Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade

15 questions
Symbiotic Relationships
Quiz
•
6th - 8th Grade

10 questions
Food Chain and Food Web
Quiz
•
8th Grade

24 questions
8th grade Science STAAR Review
Quiz
•
8th Grade