

Isotope Ion
Presentation
•
Science
•
11th Grade
•
Hard
James Gonzalez
FREE Resource
7 Slides • 30 Questions
1
Isotopes, Ions and Average Atomic Mass
By TROB

2
Ion Notation
Same as isotope
includes charge in upper right
+ means LESS ELECTRONS than PROTONS
(-) means MORE ELECTRONS than PROTONS

3
Ions
Ion means charged atom
To be charged it needs more or less electrons than protons
more electrons = NEGATIVE
less electrons = POSITIVE

4
ISOTOPES:
Same Element
Different Mass Number
WHICH MEANS DIFFERENT # of NEUTRONS

5
Average Atomic Mass
DIFFERENT than Mass Number
Average of all of the isotopes of an atom found in nature.

6
How to tell which isotope is more abundant?
CLOSEST TO ROUNDED AVERAGE ATOMIC MASS
So the most abundant isotope of Gold would be?
Gold - 197

7
Multiple Choice
8
Multiple Choice
What is the average mass of this element?
9
Multiple Choice
How many protons does this element have?
10
Multiple Choice
11
Multiple Choice
12
Multiple Choice
13
Multiple Choice
14
Multiple Choice
15
Multiple Choice

How many electrons?
9
4
5
2
16
Multiple Choice

How many neutrons?
9
4
5
2
17
Multiple Choice
18
How they get that number
% abundance of that isotope x actual mass of that isotope
add together
divide by 100 (if you didn't put into percentage first)

19
Multiple Choice

20
Multiple Choice

21
Multiple Choice

22
Multiple Choice

23
Multiple Choice
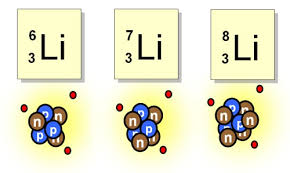
24
Multiple Choice

25
Multiple Choice

26
Multiple Choice

27
Multiple Choice

28
Multiple Choice
An atom with the same number of protons and electrons (no charge) and the expected atomic mass or number of neutrons from the periodic table.
Neutral/Normal Atom
Ion
Isotope
29
Multiple Choice
An atom that has gained or lost electrons in an attempt to become stable/bond with another atom.
Normal/Neutral Atom
Ion
Isotope
30
Multiple Choice
A negatively charged ion that has gained electrons to satisfy the octet rule (metals do this)
Anion
Cation
Neutral Atom
Isotope
31
Multiple Choice
A positively charged ion that has lost electrons to satisfy the octet rule (non-metals do this)
Anion
Cation
Neutral Atom
Isotope
32
Multiple Choice
Atoms of the same element that have different mass than expected because they have a different number of neutrons.
Neutral Atom
Ion
Isotope
Cation
33
Multiple Choice
If an atom has 9 protons and 10 electrons it has a charge of ____.
0
-1
+1
-2
34
Multiple Choice
If an atom has 4 protons and 2 electrons it has a charge of ____.
0
-2
+2
+1
35
Multiple Choice
If an atom has a charge of +3 and an atomic number of 5 it should have _______ electrons.
1
2
3
4
36
Multiple Choice
If an atom has a charge of -2 and an atomic number of 16 it should have _______ electrons.
14
16
18
20
37
Multiple Choice
If an atom has a charge of 0 (neutral) and an atomic number of 8 it should have ____ electrons.
0
8
7
9
Isotopes, Ions and Average Atomic Mass
By TROB

Show answer
Auto Play
Slide 1 / 37
SLIDE
Similar Resources on Wayground

33 questions
P.7A,B- Characteristics and Behaviors of Waves
Presentation
•
11th Grade

32 questions
Colligative properties practice
Presentation
•
10th - 12th Grade

33 questions
Motion Graphs
Presentation
•
11th Grade

32 questions
LOS COMPUESTOS ORGÁNICOS Y SU CLASIFICACIÓN
Presentation
•
11th Grade

28 questions
Solubility Curves
Presentation
•
10th - 12th Grade

28 questions
Electromagnet
Presentation
•
10th Grade

31 questions
Calvin Cycle
Presentation
•
11th Grade

33 questions
Origin of Life forms 2021
Presentation
•
11th - 12th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

46 questions
Unit 4 Geosphere Test Review
Quiz
•
9th - 12th Grade

10 questions
Explore Genetic Drift and Its Effects-March madness week 4
Quiz
•
9th - 12th Grade

20 questions
9B Fossil Record Vocabulary.S26
Quiz
•
9th - 12th Grade

6 questions
Pedigree Charts
Interactive video
•
11th Grade

13 questions
Amoeba Sisters: Biomolecules
Interactive video
•
9th - 12th Grade

20 questions
Climate Change
Quiz
•
6th - 12th Grade

10 questions
Symbiosis
Interactive video
•
11th Grade

26 questions
Adaptation and Natural Selection
Quiz
•
9th - 12th Grade