

Boyle's Law Notes
Presentation
•
Chemistry
•
9th - 12th Grade
•
Medium
Alyssa Harris
Used 66+ times
FREE Resource
9 Slides • 6 Questions
1

Page 103
2

3

4
Multiple Choice
5
Multiple Choice
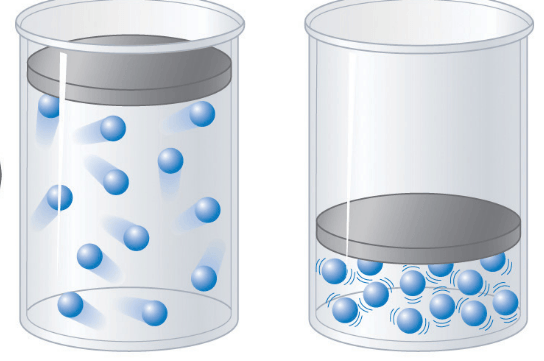
6
Multiple Choice
7

8

9

10

11

12

13
Multiple Choice
14
Multiple Choice
1. A sample of carbon dioxide occupies a volume of 3.50 liters at 125torr pressure. What pressure would the gas exert if the volume was decreased to 2.00 liters?
219 torr
222 torr
567 torr
654 torr
15
Multiple Choice
1. A sample of oxygen gas occupies a volume of 250.L at 740. torr pressure. What volume will it occupy at 800. torr pressure?
266 L
245 L
231 L
217 L

Page 103
Show answer
Auto Play
Slide 1 / 15
SLIDE
Similar Resources on Wayground

14 questions
Choosing between the passé composé and imparfait
Presentation
•
9th - 12th Grade

13 questions
Quimica 2
Presentation
•
KG

10 questions
Acids and Bases Neutralization and Titration
Presentation
•
10th - 12th Grade

12 questions
Particle/Mole Conversions
Presentation
•
9th - 12th Grade

13 questions
Calculating Molar Mass Day 1
Presentation
•
9th - 12th Grade

10 questions
Earth Sciences: Plate Boundaries
Presentation
•
9th - 12th Grade

11 questions
3.10 Solubility
Presentation
•
9th - 12th Grade

11 questions
9-1 The Mole
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

10 questions
5.P.1.3 Distance/Time Graphs
Quiz
•
5th Grade

10 questions
Fire Drill
Quiz
•
2nd - 5th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

22 questions
School Wide Vocab Group 1 Master
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Chemistry

25 questions
Solutions Test Review
Quiz
•
10th Grade

20 questions
Acids and Bases
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

15 questions
STAAR Review Quizziz 1
Quiz
•
10th Grade

15 questions
STAAR Wayground #2
Quiz
•
10th Grade

41 questions
Unit 5 Test - Biology
Quiz
•
12th Grade

15 questions
Ionic Compounds: Naming and Formula Writing
Quiz
•
9th - 12th Grade

5 questions
DOL IP: Solubility and Precipitation Reactions
Quiz
•
9th Grade