

Endothermic and Exothermic Chemical Reactions
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
14 Slides • 7 Questions
1
Content Objective: PS.3b
Learning Target: use evidence and scientific reasoning to differentiate (tell the difference) between a chemical reaction that requires an input of energy (endothermic) and one that releases energy (exothermic)
Language Objective: I can define and identify the endothermic and exothermic reactions.
2
Directions:
watch the video
write down key details and/or examples of endothermic and exothermic reactions
use the slides to write key information on the notes sheet.
answer the interactive questions

3
4
Share the key details from the video.

5
Recall:
Chemical changes involve the breaking and making of chemical bonds. Any time there is a chemical reaction, there will also be a change in energy.
Let's discuss two types of chemical change that are classified by whether the reaction absorbs or releases heat energy: endothermic reactions and exothermic reactions.

6
Multiple Choice

During chemical reactions, bonds are...
broken only
formed only
both broken and formed
none of the above are correct
7
ENDOTHERMIC REACTIONS - "endo" is a prefix that can mean "inner" or "absorbing"
If the total energy required to break bonds in the reactants is more than the total energy released when new bonds are formed in the products, the reaction is endothermic.
8
You can remember this because the prefix endo- means inner or absorbing.
So, an endothermic reaction takes thermal energy in or absorbs thermal energy from the surroundings; making the temperature of the surroundings decrease.
Energy used to break bonds > Energy released to form bonds
9
Endothermic Reaction Example
Instant cold packs are an example of an endothermic reaction that you might be familiar with. Inside, there is solid ammonium nitrate. There is also a separate bag of water. To activate the reaction, you squeeze the bag of water to cause it to break. The water is then able to mix with the ammonium nitrate and cause an endothermic reaction.

An instant cold pack contains ammonium nitrate and water. When the chemicals are mixed, it causes an endothermic reaction.
10

Remember:
An endothermic chemical reaction absorbs heat from the environment.
11
EXOTHERMIC REACTIONS - "exo" is a prefix that means "outside"
If the total energy required to break bonds in the reactants is less than the total energy released when new bonds are formed in the products, the reaction is exothermic.
12
In other words, an exothermic reaction releases heat energy into the surroundings. This causes an increase in temperature. You can remember this because the prefix exo- means outside or external.
The heat goes out of the reaction; making the temperature of the surroundings increase.
Energy used to break bonds < Energy released to form bonds
13
Exothermic Reaction Example
Burning the wax of a candle is an example of an exothermic reaction because heat from the flame is released to the environment.

14

Remember:
An exothermic chemical reaction releases heat to the environment.
15
Multiple Choice
If a chemical reaction is EXOTHERMIC, the temperature of the surrounding would....
stay the same
increase
decrease
16
Multiple Choice
During an endothermic reaction in a beaker if we are part of the surroundings and touched the beaker, it would feel _______.
warm
cold
17
Multiple Choice
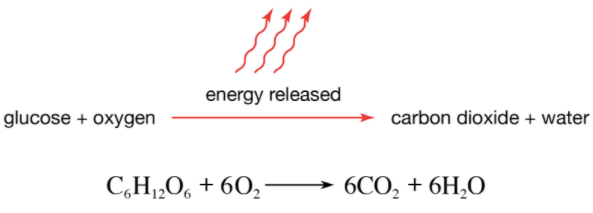
The illustration shows cell respiration. Is the overall reaction of cell respiration an Endothermic or an Exothermic reaction?
endothermic
exothermic
18
Multiple Choice

A student mixed two chemicals to allow them to react. The temperature before the reaction was 25 ° C. The temperature after the reaction was 18° C. Which of the following is true?
The temperature did not change.
It is an endothermic reaction
It is an exothermic reaction
Energy is released from the reaction into the surrounding.
19
Multiple Choice

What type of reaction occurs in a hand warmer?
exothermic
endothermic
20
Multiple Choice

What instrument would you use to detect an endothermic or exothermic reaction?
triple beam balance
ruler
thermometer
21
Glue your notes in your notebook, please.

Content Objective: PS.3b
Learning Target: use evidence and scientific reasoning to differentiate (tell the difference) between a chemical reaction that requires an input of energy (endothermic) and one that releases energy (exothermic)
Language Objective: I can define and identify the endothermic and exothermic reactions.
Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

16 questions
Speed/Time Graphs
Presentation
•
8th Grade

16 questions
Tides
Presentation
•
8th Grade

17 questions
Genetics Review Part 2 (Practice)
Presentation
•
6th - 9th Grade

15 questions
Cell and cell organelle
Presentation
•
8th Grade

17 questions
Motors and Generators
Presentation
•
8th Grade

16 questions
Sci 8 - 2.1 Microscopes
Presentation
•
8th Grade

18 questions
Cellular respiration vs photosynthesis
Presentation
•
8th Grade

14 questions
Combustion & Flame
Presentation
•
8th Grade
Popular Resources on Wayground

20 questions
STAAR Review Quiz #3
Quiz
•
8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

6 questions
Marshmallow Farm Quiz
Quiz
•
2nd - 5th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

45 questions
Food Webs
Quiz
•
6th - 8th Grade

50 questions
Stars, Galaxies, HR Diagram
Quiz
•
8th Grade

40 questions
Unit F1 Review
Quiz
•
8th Grade

20 questions
Energy Transformations
Quiz
•
6th - 8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade