

Parts of a Chemical Equation
Presentation
•
Science
•
8th Grade
•
Hard
Joseph Anderson
FREE Resource
10 Slides • 15 Questions
1
Parts of a Chemical Equation
An overview starting with Chemical Symbols and working up to Chemical Formulas

2
What is a chemical formula?
A chemical formula is a way to represent a chemical reaction through the use of NUMBERS and CHEMICAL SYMBOLS
3
To understand a chemical formula you must...
Know how to read the chemical symbols
know how to read the periodic table
Know when to multiply numbers
When to add numbers
4
Reading Chemical Symbols
To read a chemical symbol, you first, need to know what a chemical symbol is.
A chemical symbol is defined as a single capital letter or one capital and one lower case letter.
This means that when you are reading chemical symbols, if you see two or more capital letters side-by-side, that each capital letter represents a different element.
5
Multiple Select
Select all of the following examples that contain at least two elements based on the chemical symbols shown:
H2+O= H2O
C + H4= CH4
O3+O2=O5
Q3+Cl2= 6QCL
6
To Read a Chemical Formula
You have to make sure you know how to read the Periodic Table of Elements. This table shows you every element that has been discovered so far, and it also shows the chemical symbol for each element.
7
Fill in the Blanks
Type answer...
8
Reading the numbers...
When looking at a chemical formula, you will see two types of numbers: a coefficient or a subscript.
9
Open Ended
Can you tell me what a coefficient is?
10
Coefficients
Are the LARGE NUMBERS located to the left of a chemical symbol.
An important thing to remember is that EVERY SYMBOL behind a coefficient gets multiplied by that coefficient.
11
Multiple Choice
Which of the following choices shows the correct number of atoms present in molecule 5NaHCO3?
8 atoms
15 atoms
23 atoms
30 atoms
12
Fill in the Blanks
Type answer...
13
You will multiply to count atoms when
You see a coefficient
You see a coefficient and a subscript
You see parenthesis and then a subscript
You see a subscript inside the parenthesis and then a subscript outside of the parenthesis
14
You will add when counting atoms if
You see the same element listed multiple times in the chemical formula
15
Open Ended
In your opinion, what is a subscript?
16
A subscript is the number
To the lower right of the chemical symbol that represents the number of atoms in a single molecule of an atom.
17
Multiple Choice
How many Aluminum atoms are in Al2O3?
3
2
5
18
Multiple Choice
Al2
A single atom
Elemental Molecule
Compound Molecule
None of these
19
Multiple Choice
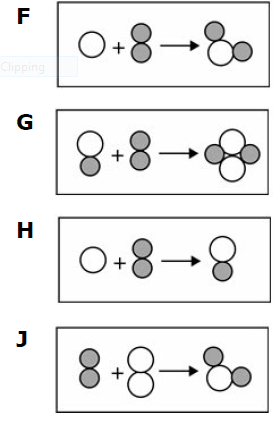
Which of the following models best demonstrates a balanced chemical equation?
F
G
H
J
20
Multiple Choice
Which of the following chemical equations follows the Law of Conservation of Mass? Hint: which is balanced?
CO2 + H2O --> C6H12O6 + O2
C6O + H12O --> C6H12O2
CO2 + H2O --> C6H12O6 + O2
CO2 +H2O --> C6H12O6 +O2
21
Multiple Choice

How many atoms are represented in the reactants of this equation?
6
12
24
36
22
Multiple Choice
2H2O + 2N → 4H + 2NO
The chemical symbols on the RIGHT (at the end of the equation) are called:
products
reactants
yields/produces
23
Multiple Choice
In the equation, 2Mg + O2 ---> 2MgO, which are the reactants?
2Mg and O2
2MgO
Mg and MgO
O and MgO
24
Multiple Choice
How many elements are in C6H12O6?
1
2
3
4
25
Multiple Choice
three hydrogen (H)
one sulfur (S)
four oxygen (O)
Parts of a Chemical Equation
An overview starting with Chemical Symbols and working up to Chemical Formulas

Show answer
Auto Play
Slide 1 / 25
SLIDE
Similar Resources on Wayground

20 questions
Science Tools
Presentation
•
8th Grade

20 questions
Cluster 4 Day 1-Symbiosis
Presentation
•
8th Grade

19 questions
Geologic Time Review
Presentation
•
8th Grade

19 questions
Review- Electromagnetism
Presentation
•
8th Grade

18 questions
Plant Reproduction
Presentation
•
8th Grade

18 questions
Lesson 5.5: The Theory of Plate Tectonics
Presentation
•
8th Grade

21 questions
Earth Layers
Presentation
•
8th Grade

19 questions
Aquatic Ecosystem & Ocean zones
Presentation
•
8th Grade
Popular Resources on Wayground

10 questions
Fire Safety Quiz
Quiz
•
12th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

36 questions
6th Grade Math STAAR Review
Quiz
•
6th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade

12 questions
What makes Nebraska's government unique?
Quiz
•
4th - 5th Grade
Discover more resources for Science

20 questions
8th Grade Science STAAR Review
Quiz
•
8th Grade

8 questions
Amoeba Sister Asexual vs Sexual Reproduction
Interactive video
•
8th Grade

19 questions
Introduction to Properties of Waves
Presentation
•
6th - 8th Grade

16 questions
Interactions within Ecosystems
Presentation
•
6th - 8th Grade

68 questions
8th Grade Physical Science GMAS Challenge
Quiz
•
8th Grade

100 questions
GMAS Science Review
Quiz
•
8th Grade

10 questions
Newton's Laws of Motion
Quiz
•
8th Grade

46 questions
8th Science STAAR Review
Quiz
•
8th Grade