

Rate of Reactions
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
8 Slides • 13 Questions
1
Reaction rates
2

3

4

5
Multiple Choice
true
false
6
Multiple Choice
The energy needed for a reaction to occur
The minimum amount of energy needed for a reaction to potentially occur
The energy added by a catalyst
The energy possessed by the products
7
Multiple Choice
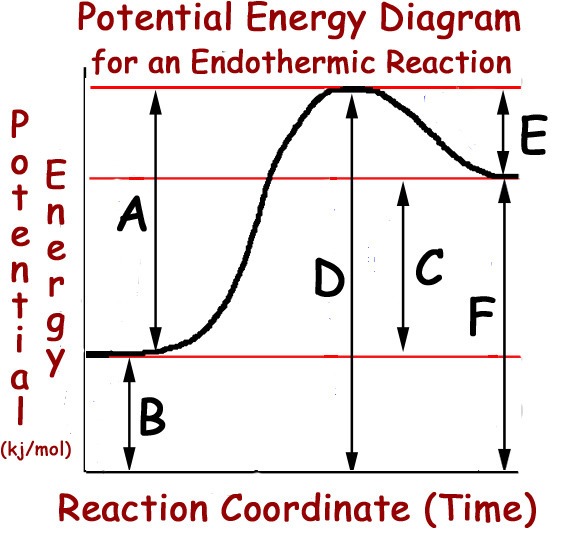
A
B
C
D
8
Multiple Choice
What is collision theory?
Molecules must collide in the correct orientation with enough energy to bond.
Molecules need enough energy to collide and react.
Atoms constantly collide and react.
The minimum energy needed for atoms to react
9
Multiple Choice
True
False
10
Multiple Choice
What is the rate of reaction?
How much energy is needed for a reaction to occur.
The energy required to break a bond.
The time it takes for a reaction to occur.
Collision Theory
11
12

13

14

15
Multiple Choice
What is the purpose of a catalyst?
Helps to slow down a reaction
Raises the activation energy
Lowers the activation energy
Is consumed by the reaction
16
Multiple Choice
What factors effect rate of reaction?
Temperature, Concentration, Pressure and Energy
Surface Area, Concentration, Energy and Pressure
Temperature, Pressure, Concentration and Surface area
Pressure, Surface area, Density and Energy
17
Multiple Choice
Increasing the temperature of your solution will.......
Not affect the rate of reaction.
Speed up the rate of reaction
Slow down the rate of reaction
18
Multiple Choice
Slow collision frequency
Allow less effective collision between the particles
Cause particles to lose speed
Increase collision between the particles thus increasing the rate.
19
Multiple Choice
concentration
surface area
temperature
reactants
20
Multiple Choice
catalyst
product
reactant
solute
21
Multiple Choice
it increases the amount of reactants
it lowers the activation energy
it increases the energy of particle collisions
it increases the frequency of particle collisions because there are more collision sites
Reaction rates
Show answer
Auto Play
Slide 1 / 21
SLIDE
Similar Resources on Wayground

17 questions
Gas Laws - Dalton's Partial Pressure
Lesson
•
9th - 12th Grade

17 questions
Nuclear Decay
Lesson
•
9th - 12th Grade

18 questions
Quantitative vs Qualitative observations
Lesson
•
9th - 12th Grade

14 questions
Electron Math
Lesson
•
9th - 12th Grade

16 questions
Scientific Method
Lesson
•
9th - 11th Grade

14 questions
L1 Types of Matter
Lesson
•
10th - 12th Grade

16 questions
Cell Cycle and Cancer Lesson
Lesson
•
9th - 12th Grade
Popular Resources on Wayground

15 questions
Fractions on a Number Line
Quiz
•
3rd Grade

14 questions
Boundaries & Healthy Relationships
Lesson
•
6th - 8th Grade

13 questions
SMS Cafeteria Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Equivalent Fractions
Quiz
•
3rd Grade

25 questions
Multiplication Facts
Quiz
•
5th Grade

12 questions
SMS Restroom Expectations Quiz
Quiz
•
6th - 8th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

10 questions
Pi Day Trivia!
Quiz
•
6th - 9th Grade
Discover more resources for Chemistry

13 questions
Solubility Curves
Quiz
•
10th Grade

10 questions
Exploring Nuclear Fission and Fusion
Interactive video
•
6th - 10th Grade

5 questions
Exit Check 4.1 - Balancing Chemical Equations
Quiz
•
10th Grade

20 questions
Predicting Products
Quiz
•
9th - 12th Grade

8 questions
Labeling a Reaction
Quiz
•
9th - 12th Grade

92 questions
Chem Quarter 3 Review
Quiz
•
10th Grade

19 questions
Isotopes & Ions
Quiz
•
9th Grade

20 questions
Naming & Writing Chemical Formulas
Quiz
•
10th Grade