

Gas Laws
Presentation
•
Chemistry
•
9th - 12th Grade
•
Hard
Joseph Anderson
FREE Resource
7 Slides • 11 Questions
1
Gas Laws Lesson - Part 3
By Michaela Bratsch
2

3

4
Multiple Choice
5
Multiple Choice
6
Multiple Choice
What does R stand for
Ideal gas constant
Real gas constant
Temperature constant
Ideal gas law
7
Multiple Choice
Which is a correct unit for R?
L.atm/mol.k
L.atm
mmHg/mol.K
L.atom/mol
8
Multiple Choice
The ideal gas law is an equation that relates which variables to a constant of R?
9
Multiple Choice
10

11

12

13

14
Multiple Choice
Which equation represents Dalton's Law?
V1 / T1 = V2 / T2
PV = nRT
P1 / T1 = P2 / T2
Ptotal = P1 + P2 + P3
15
Multiple Choice
The amount of force pushing on an area is called....
Temperature
Viscosity
Volume
Pressure
16
Multiple Choice
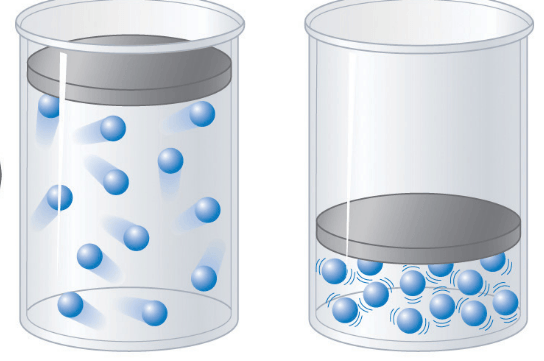
Which container will have a lower pressure?
left
right
they both have the same pressure
17
Multiple Choice
A mixture of H, Ne, & O has a pressure of 12.4 atm. If Hydrogen measures 5.6 atm and Oxygen measures 2.0 atm, what is the pressure of Neon?
7.6 L
4.8 L
5.4 L
12.0 L
18
Multiple Choice
CO2, with a pressure of 124 kPa, and Ar, with a pressure of 98.0 kPa are mixed into a container. If 7 liters of Cl2 with a pressure of 115 kPa is then added to the mixture, what is the total kPa?
224 kPa
337 kPa
107 kPa
1027 kPa
Gas Laws Lesson - Part 3
By Michaela Bratsch
Show answer
Auto Play
Slide 1 / 18
SLIDE
Similar Resources on Wayground

10 questions
Calculating heat of phase changes
Presentation
•
9th - 12th Grade

13 questions
Accuracy vs. Precision
Presentation
•
10th - 12th Grade

13 questions
Charles' Law Notes
Presentation
•
9th - 12th Grade

15 questions
Belajar Kimia dari Rumah
Presentation
•
9th - 12th Grade

12 questions
Structure of the Atom
Presentation
•
9th - 12th Grade

13 questions
Ch. 3.1 Measurements & Their Uncertainty - Part 1
Presentation
•
9th - 11th Grade

11 questions
3 Periodic Table
Presentation
•
9th - 12th Grade

13 questions
Debit vs Credit Cards
Presentation
•
9th - 12th Grade
Popular Resources on Wayground

19 questions
Naming Polygons
Quiz
•
3rd Grade

10 questions
Prime Factorization
Quiz
•
6th Grade

20 questions
Math Review
Quiz
•
3rd Grade

15 questions
Fast food
Quiz
•
7th Grade

20 questions
Main Idea and Details
Quiz
•
5th Grade

20 questions
Context Clues
Quiz
•
6th Grade

20 questions
Inferences
Quiz
•
4th Grade

19 questions
Classifying Quadrilaterals
Quiz
•
3rd Grade
Discover more resources for Chemistry

34 questions
Unit 11 Acids and Bases Test Review
Quiz
•
10th Grade

20 questions
Types of Chemical Reactions
Quiz
•
9th - 12th Grade

120 questions
2nd Semester Final Review
Quiz
•
11th Grade

25 questions
Unit 12 Thermochemistry Review
Quiz
•
10th Grade

22 questions
Solubility Curve Practice
Quiz
•
10th Grade

25 questions
Solutions Test Review
Quiz
•
10th Grade

12 questions
IP PreAP: Fission and Fusion Reactions
Quiz
•
10th - 12th Grade

22 questions
Unit 8- Chemical Reactions
Quiz
•
10th Grade